Edexcel iGCSE Chemistry -3.9 Factors Affecting Rate of Reaction- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -3.9 Factors Affecting Rate of Reaction- Study Notes- New syllabus
Edexcel iGCSE Chemistry -3.9 Factors Affecting Rate of Reaction- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
3.9 describe experiments to investigate the effects of changes in surface area of a solid, concentration of a solution, temperature and the use of a catalyst on the rate of a reaction
3.9 Investigating Factors Affecting the Rate of Reaction
Definition:
The rate of reaction is the speed at which reactants are converted into products.

Rate can be measured by:
- Volume of gas produced over time.
- Loss of mass over time.
- Time taken for a visible change (e.g. colour change or precipitate).
1. Effect of Surface Area
Example Reaction:
\( \mathrm{CaCO_3(s) + 2HCl(aq) \rightarrow CaCl_2(aq) + CO_2(g) + H_2O(l)} \)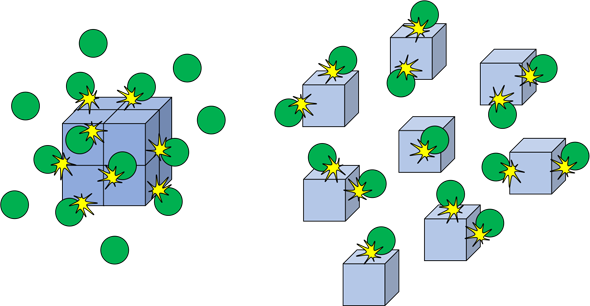
Method:
- Use equal masses of calcium carbonate chips and powder.
- Add each to the same volume and concentration of hydrochloric acid.
- Measure volume of \( \mathrm{CO_2} \) produced over time.
Observation:
- Powder reacts faster than chips.
Explanation:
- Greater surface area exposed.
- More frequent successful collisions.
2. Effect of Concentration
Example Reaction:
\( \mathrm{Na_2S_2O_3(aq) + 2HCl(aq) \rightarrow 2NaCl(aq) + SO_2(g) + S(s) + H_2O(l)} \)
Method (Disappearing Cross Experiment):
- Place sodium thiosulfate solution in a flask over a paper cross.
- Add hydrochloric acid.
- Measure time taken for cross to disappear (due to sulfur precipitate).
- Repeat with different concentrations.
Observation:
- Higher concentration → shorter time → faster rate.
Explanation:
- More particles per unit volume.
- More frequent collisions.
3. Effect of Temperature
Can be investigated using the same reaction as above.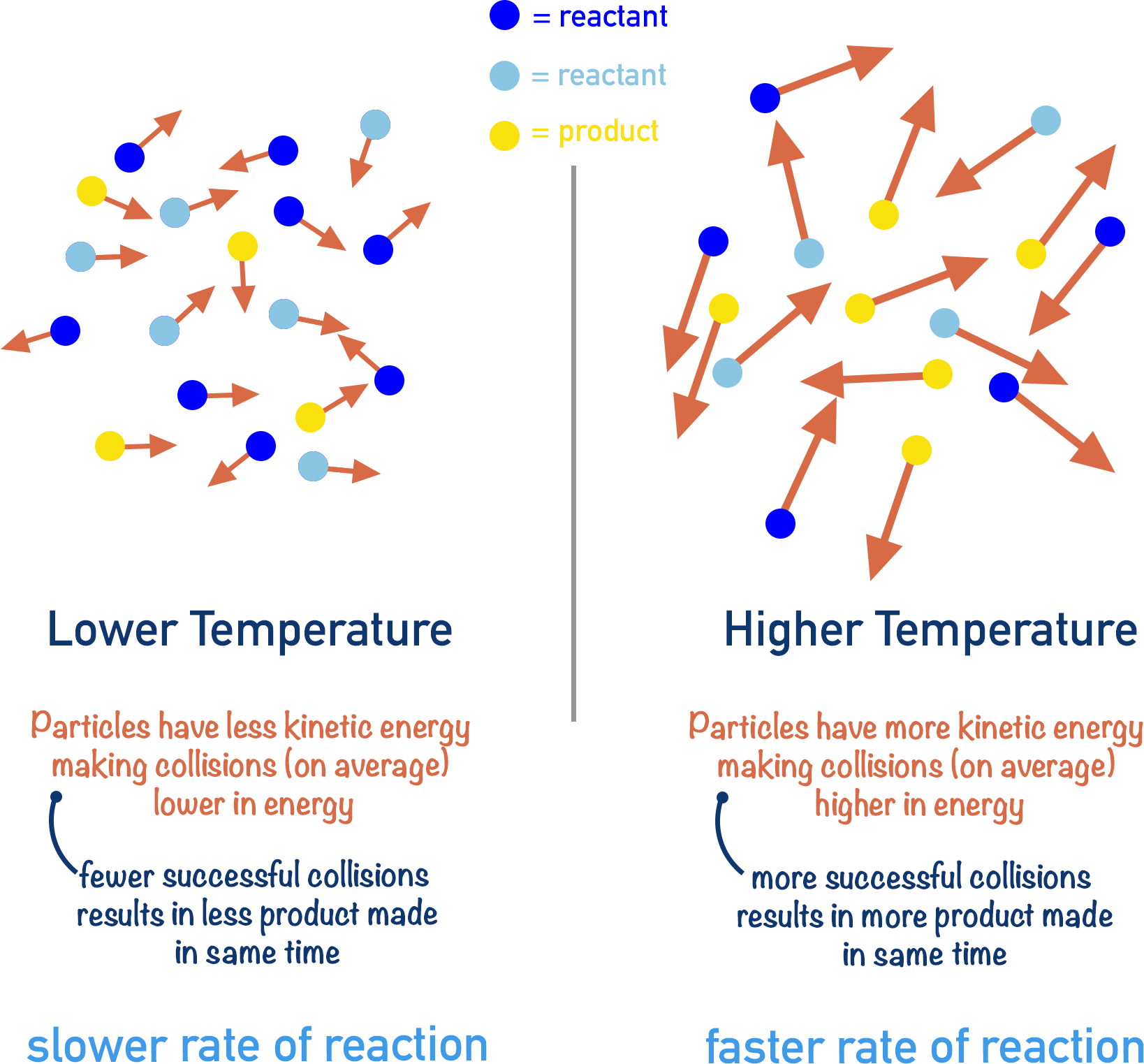
Method:
- Warm the sodium thiosulfate solution to different temperatures.
- Add acid and measure time for cross to disappear.
Observation:
- Higher temperature → faster reaction.
Explanation:
- Particles have more kinetic energy.
- Collisions occur more frequently.
- More collisions exceed activation energy.
4. Effect of a Catalyst
Example Reaction:
Decomposition of hydrogen peroxide:
\( \mathrm{2H_2O_2(aq) \rightarrow 2H_2O(l) + O_2(g)} \)![]()
Method:
- Add manganese dioxide as a catalyst.
- Measure oxygen produced over time.
Observation:
- Reaction is much faster with catalyst.
Explanation:
- Catalyst provides an alternative pathway.
- Lowers activation energy.
- More successful collisions.
- Catalyst is not used up.
| Factor | What Changes | Effect on Rate |
|---|---|---|
| Surface area | More exposed particles | Increases |
| Concentration | More particles per volume | Increases |
| Temperature | Higher kinetic energy | Increases |
| Catalyst | Lower activation energy | Increases |
Example 1 (Conceptual):
Why does powdered calcium carbonate react faster than marble chips?
▶️ Answer/Explanation
Powder has larger surface area.
More particles are exposed.
Collisions occur more frequently.
Example 2 (Application):
If the concentration of acid is doubled, what happens to the reaction rate?
▶️ Answer/Explanation
More particles per unit volume.
More frequent collisions.
Rate increases.
Example 3 (Hard ):
Explain fully, using collision theory, why increasing temperature increases reaction rate.
▶️ Answer/Explanation
Particles gain kinetic energy.
They move faster.
Collisions occur more frequently.
A greater proportion of collisions have energy greater than activation energy.
Therefore more successful collisions occur.
The reaction rate increases.
