Edexcel iGCSE Chemistry -4.1 Hydrocarbons- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.1 Hydrocarbons- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.1 Hydrocarbons- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.1 know that a hydrocarbon is a compound of hydrogen and carbon only
4.1 Hydrocarbons
A hydrocarbon is a compound that contains only hydrogen and carbon atoms.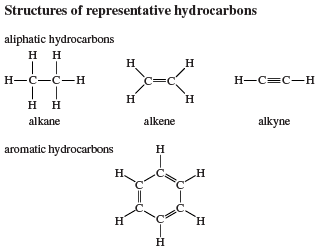
There are no other elements present.
General Characteristics
- Contain only \( \mathrm{C} \) and \( \mathrm{H} \)
- Usually found in crude oil and natural gas
- Often used as fuels
- Undergo combustion reactions
Examples of Hydrocarbons
Methane:
\( \mathrm{CH_4} \)
Ethane:
\( \mathrm{C_2H_6} \)
Ethene:
\( \mathrm{C_2H_4} \)
Combustion of Hydrocarbons
When hydrocarbons burn completely in oxygen, they produce carbon dioxide and water.

\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Incomplete combustion can produce carbon monoxide or carbon (soot).
Why Hydrocarbons Are Important
- Main components of fossil fuels
- Used for heating and electricity
- Raw materials for making plastics and other chemicals
| Compound | Contains Only C and H? | Hydrocarbon? |
|---|---|---|
| \( \mathrm{CH_4} \) | Yes | Yes |
| \( \mathrm{C_2H_5OH} \) | No (contains O) | No |
| \( \mathrm{C_2H_6} \) | Yes | Yes |
Example 1 (Conceptual):
Why is \( \mathrm{C_2H_5OH} \) not a hydrocarbon?
▶️ Answer/Explanation
Because it contains oxygen as well as carbon and hydrogen.
A hydrocarbon must contain only carbon and hydrogen.
Example 2 (Application):
Is \( \mathrm{C_3H_8} \) a hydrocarbon? Explain.
▶️ Answer/Explanation
Yes.
It contains only carbon and hydrogen atoms.
Example 3 (Hard):
A compound has the formula \( \mathrm{C_4H_{10}} \). Explain fully why this compound is classified as a hydrocarbon and describe what happens when it burns in oxygen.
▶️ Answer/Explanation
The formula \( \mathrm{C_4H_{10}} \) shows the compound contains only carbon and hydrogen.
Therefore it is a hydrocarbon.
During complete combustion it reacts with oxygen.
The products formed are carbon dioxide and water.
This reaction releases heat energy.
