Edexcel iGCSE Chemistry -4.10 Properties of Fractions- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.10 Properties of Fractions- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.10 Properties of Fractions- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.10 know the trend in colour, boiling point and viscosity of the main fractions
4.10 Trends in Colour, Boiling Point and Viscosity of the Main Fractions
The fractions obtained from crude oil show clear trends as you move from the top to the bottom of the fractionating column.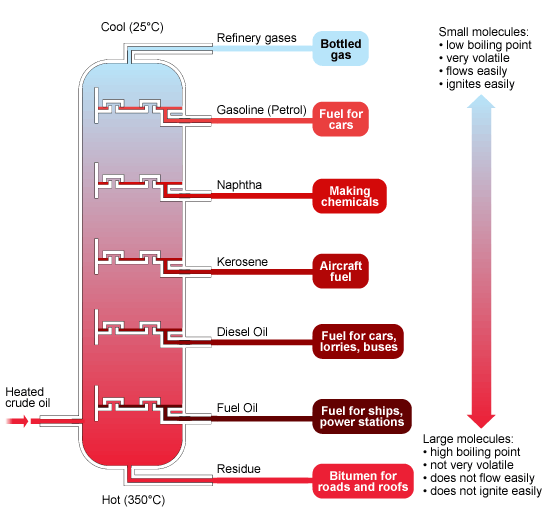
These trends are due to increasing hydrocarbon chain length.
Trend in Boiling Point
- Boiling point increases down the column.
- Small molecules → low boiling point.
- Large molecules → high boiling point.
Larger molecules have stronger intermolecular forces, so more energy is needed to separate them.
Trend in Viscosity
Viscosity means how thick a liquid is.
- Viscosity increases down the column.
- Small molecules → runny (low viscosity).
- Large molecules → thick and sticky (high viscosity).
Trend in Colour
- Fractions become darker down the column.
- Top fractions are colourless or pale.
- Bottom fractions are dark brown or black.
| Position in Column | Boiling Point | Viscosity | Colour |
|---|---|---|---|
| Top | Low | Low (runny) | Colourless / pale |
| Middle | Medium | Moderate | Yellow to light brown |
| Bottom | High | High (thick) | Dark brown / black |
Key Reason for the Trends
- Chain length increases down the column.
- Intermolecular forces increase.
- Boiling point increases.
- Viscosity increases.
- Colour darkens.
Example 1 (Conceptual):
Why do larger hydrocarbons have higher boiling points?
▶️ Answer/Explanation
They have stronger intermolecular forces.
More energy is required to overcome these forces.
Example 2 (Application):
Which fraction would be most viscous?
▶️ Answer/Explanation
The fraction collected at the bottom (e.g. bitumen).
It contains very long hydrocarbon chains.
Example 3 (Hard ):
Explain fully the trends in boiling point, viscosity and colour as you move down the fractionating column.
▶️ Answer/Explanation
Moving down the column, hydrocarbon chain length increases.
Larger molecules have stronger intermolecular forces.
Therefore more energy is needed to separate the molecules, so boiling point increases.
Longer chains become more tangled, increasing viscosity.
The fractions also become darker in colour.
Thus boiling point and viscosity increase and colour darkens down the column.
