Edexcel iGCSE Chemistry -4.11 Fuels and Energy Release- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.11 Fuels and Energy Release- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.11 Fuels and Energy Release- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.11 know that a fuel is a substance that, when burned, releases heat energy
4.11 Fuels
A fuel is a substance that, when burned, releases heat energy.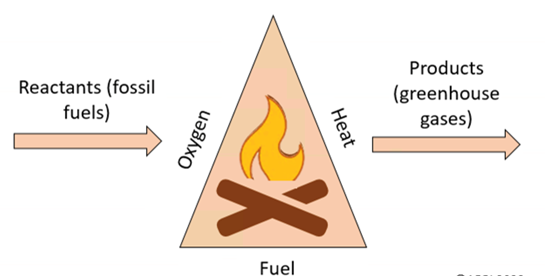
Burning is a combustion reaction with oxygen.
Definition
A fuel is a substance that reacts with oxygen to release energy in the form of heat.
Combustion Reaction
Most fuels are hydrocarbons.
Complete combustion produces carbon dioxide and water.
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Energy is released as heat (exothermic reaction).
Examples of Fuels
![]()
- Coal
- Petrol (gasoline)
- Diesel
- Natural gas (methane)
- Hydrogen
Uses of Fuels
- Heating homes
- Cooking
- Generating electricity
- Powering vehicles
| Fuel | Main Product of Complete Combustion | Energy Released? |
|---|---|---|
| Methane | \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \) | Yes |
| Petrol | \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \) | Yes |
| Hydrogen | \( \mathrm{H_2O} \) | Yes |
Key GCSE Point
- Fuel → releases energy when burned.
- Combustion → exothermic reaction.
- Energy released is usually used for heating or work.
Example 1 (Conceptual):
Why is combustion of fuels described as exothermic?
▶️ Answer/Explanation
Because heat energy is released to the surroundings.
Example 2 (Application):
State one reason why methane is used as a fuel.
▶️ Answer/Explanation
It releases heat energy when burned.
Example 3 (Hard):
Explain fully why hydrogen can be described as a fuel.
▶️ Answer/Explanation
Hydrogen reacts with oxygen in a combustion reaction.
The reaction releases heat energy.
The equation is:
\( \mathrm{2H_2 + O_2 \rightarrow 2H_2O} \)
The energy released can be used for heating or to generate electricity.
Therefore hydrogen is a fuel because it releases energy when burned.
