Edexcel iGCSE Chemistry -4.12 Combustion of Hydrocarbons- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.12 Combustion of Hydrocarbons- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.12 Combustion of Hydrocarbons- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.12 know the possible products of complete and incomplete combustion of hydrocarbons with oxygen in the air
4.12 Complete and Incomplete Combustion of Hydrocarbons
Hydrocarbons react with oxygen in the air in combustion reactions.
The products formed depend on whether there is a sufficient supply of oxygen.
1. Complete Combustion
Occurs when there is plenty of oxygen.
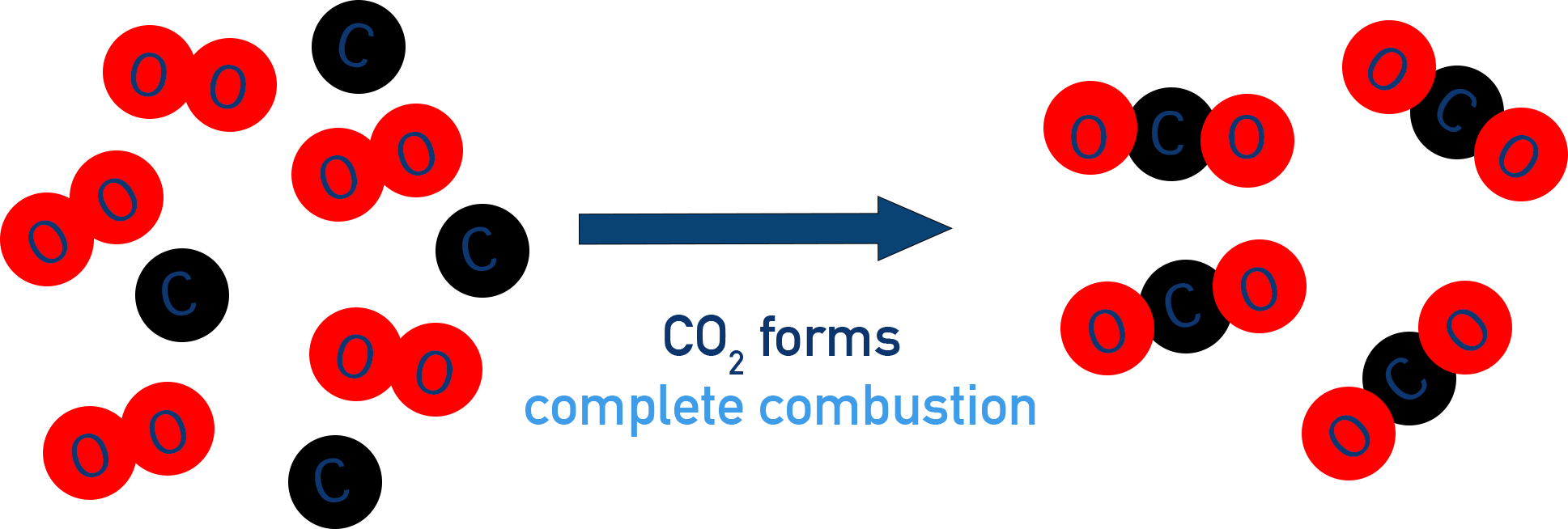
Products of complete combustion:
- Carbon dioxide \( \mathrm{CO_2} \)
- Water \( \mathrm{H_2O} \)
Example (methane):
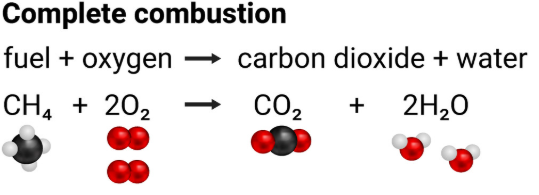
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Complete combustion produces the maximum amount of energy.
2. Incomplete Combustion
Occurs when there is limited oxygen.

Possible products:
- Carbon monoxide \( \mathrm{CO} \)
- Carbon (soot) \( \mathrm{C} \)
- Water \( \mathrm{H_2O} \)
Example producing carbon monoxide:
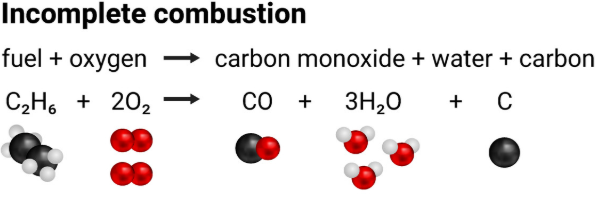
\( \mathrm{2CH_4 + 3O_2 \rightarrow 2CO + 4H_2O} \)
Example producing carbon (soot):
\( \mathrm{CH_4 + O_2 \rightarrow C + 2H_2O} \)
Why Incomplete Combustion is Dangerous
- Carbon monoxide is toxic.
- It is colourless and odourless.
- It reduces the blood’s ability to carry oxygen.
- Soot contributes to air pollution and respiratory problems.
| Type of Combustion | Oxygen Supply | Products |
|---|---|---|
| Complete | Excess oxygen | \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \) |
| Incomplete | Limited oxygen | \( \mathrm{CO} \), \( \mathrm{C} \), \( \mathrm{H_2O} \) |
Exam Key Words
- Complete combustion → carbon dioxide + water.
- Incomplete combustion → carbon monoxide and/or carbon.
- Depends on oxygen supply.
Example 1 (Conceptual):
Why does incomplete combustion occur?
▶️ Answer/Explanation
Because there is not enough oxygen for complete combustion.
Example 2 (Application):
State one product of incomplete combustion that is toxic.
▶️ Answer/Explanation
Carbon monoxide \( \mathrm{CO} \).
Example 3 (Hard ):
Explain fully the difference between complete and incomplete combustion of a hydrocarbon.
▶️ Answer/Explanation
Complete combustion occurs when there is excess oxygen.
It produces carbon dioxide and water.
Incomplete combustion occurs when oxygen is limited.
It produces carbon monoxide and/or carbon as well as water.
Carbon monoxide is toxic.
The difference depends on the amount of oxygen available.
