Edexcel iGCSE Chemistry -4.14 Oxides of Nitrogen in Engines- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.14 Oxides of Nitrogen in Engines- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.14 Oxides of Nitrogen in Engines- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.14 know that, in car engines, the temperature reached is high enough to allow nitrogen and oxygen from air to react, forming oxides of nitrogen
4.14 Formation of Oxides of Nitrogen in Car Engines
Air is mainly a mixture of nitrogen \( \mathrm{N_2} \) (about 78%) and oxygen \( \mathrm{O_2} \) (about 21%).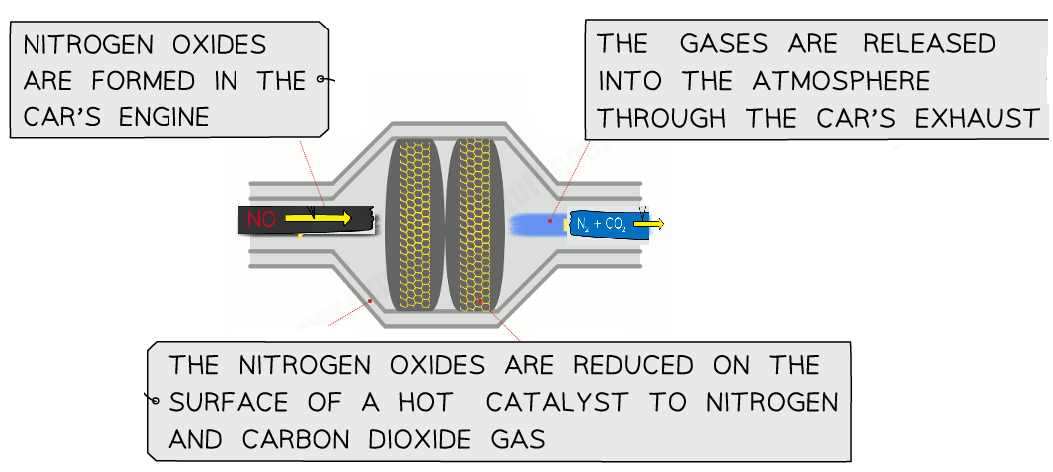
Under normal conditions, nitrogen is relatively unreactive.
High Temperatures in Car Engines
In a car engine:
- Fuel combusts at very high temperatures.
- Temperatures are high enough to overcome the strong bond in \( \mathrm{N_2} \).
- Nitrogen and oxygen react together.
Formation of Oxides of Nitrogen (NOx)
At high temperatures:
\( \mathrm{N_2 + O_2 \rightarrow 2NO} \)
Nitric oxide \( \mathrm{NO} \) can further react with oxygen:
\( \mathrm{2NO + O_2 \rightarrow 2NO_2} \)
These gases are called oxides of nitrogen (often written as NOx).
Why This Only Happens at High Temperatures
- Nitrogen has a very strong triple bond.
- Large amounts of energy are needed to break this bond.
- The high temperature inside an engine provides this energy.
Environmental Impact
- Contribute to acid rain.
- Contribute to air pollution.
- Can irritate lungs and cause breathing problems.
| Reactants | Condition | Products |
|---|---|---|
| \( \mathrm{N_2} \) and \( \mathrm{O_2} \) | Very high temperature (engine) | \( \mathrm{NO} \) and \( \mathrm{NO_2} \) |
Exam Focus
- Oxides of nitrogen form due to high engine temperatures.
- They are not from the fuel itself.
- They are formed from nitrogen and oxygen in the air.
Example 1 (Conceptual):
Why does nitrogen normally not react with oxygen in the air?
▶️ Answer/Explanation
The nitrogen molecule has a strong triple bond.
At normal temperatures there is not enough energy to break it.
Example 2 (Application):
Explain why oxides of nitrogen are produced in car engines but not in everyday air.
▶️ Answer/Explanation
Car engines reach very high temperatures.
This provides enough energy for nitrogen and oxygen to react.
In normal air, the temperature is too low.
Example 3 (Hard ):
Explain fully how oxides of nitrogen are formed in car engines and why they are harmful.
▶️ Answer/Explanation
Inside a car engine, fuel combusts at very high temperatures.
The high temperature provides enough energy to break the strong bond in nitrogen molecules.
Nitrogen reacts with oxygen from the air to form nitric oxide \( \mathrm{NO} \).
Further reactions can form nitrogen dioxide \( \mathrm{NO_2} \).
These gases are called oxides of nitrogen.
They contribute to air pollution and acid rain.
They can also irritate the respiratory system.
