Edexcel iGCSE Chemistry -4.15 Sulfur Dioxide from Impurities- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.15 Sulfur Dioxide from Impurities- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.15 Sulfur Dioxide from Impurities- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.15 explain how the combustion of some impurities in hydrocarbon fuels results in the formation of sulfur dioxide
4.15 Formation of Sulfur Dioxide from Impurities in Fuels
Hydrocarbon fuels are mainly compounds of carbon and hydrogen.
However, some fuels contain small amounts of sulfur impurities.
What Happens During Combustion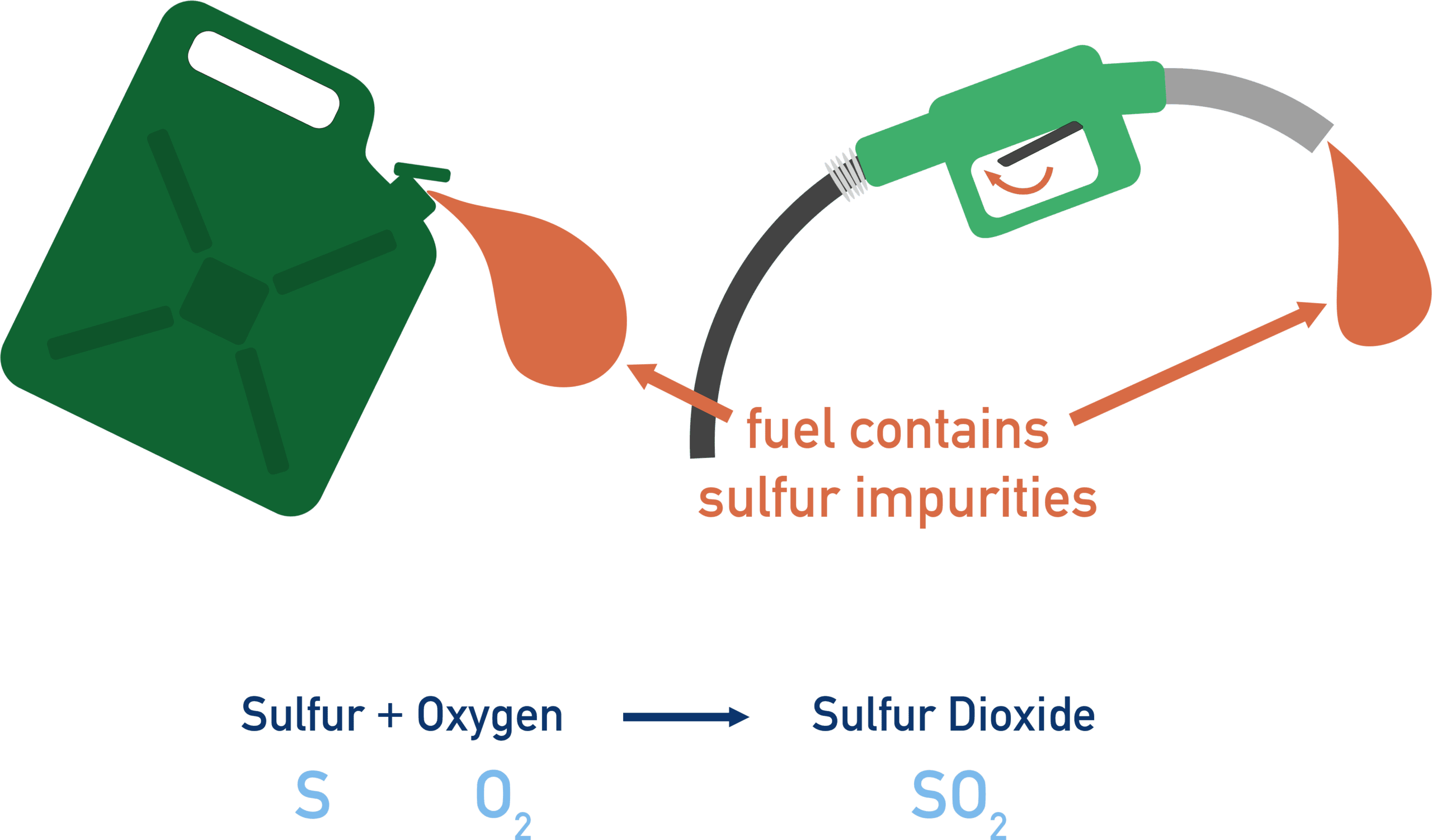
When the fuel burns:
- The hydrocarbon reacts with oxygen.
- The sulfur impurities also react with oxygen.
Sulfur is oxidised to form sulfur dioxide.
\( \mathrm{S + O_2 \rightarrow SO_2} \)
Why This Happens
- Combustion provides heat energy.

- Oxygen is present in excess air.
- Sulfur reacts readily with oxygen at high temperatures.
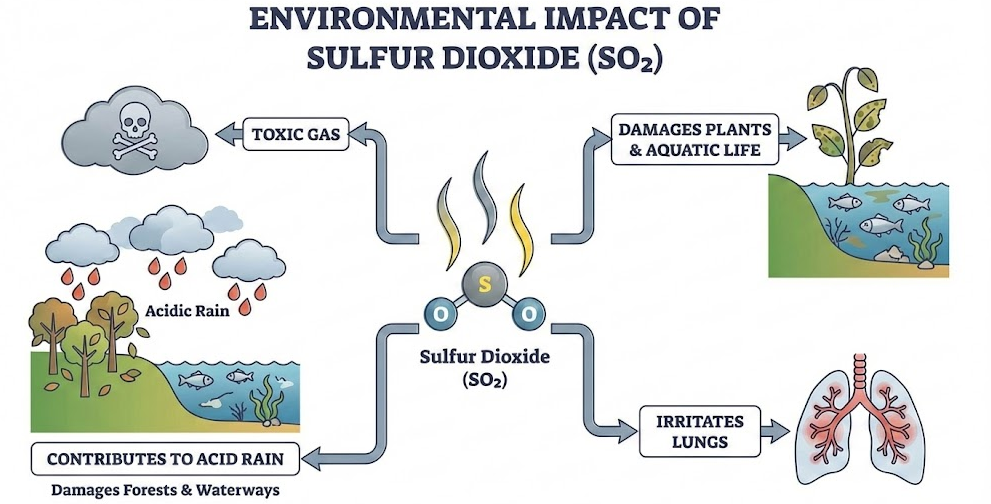
Environmental Impact
Sulfur dioxide \( \mathrm{SO_2} \):
- Is a toxic gas.
- Contributes to acid rain.
- Can damage plants and aquatic life.
- Can irritate lungs.
| Fuel Contains | During Combustion | Product Formed |
|---|---|---|
| Sulfur impurity | Reacts with oxygen | \( \mathrm{SO_2} \) |
Exam Focus
- Sulfur is an impurity in some fuels.
- When burned, sulfur reacts with oxygen.
- This forms sulfur dioxide.
- Sulfur dioxide causes acid rain.
Example 1 (Conceptual):
Why is sulfur dioxide produced when some fuels burn?
▶️ Answer/Explanation
The fuel contains sulfur impurities.
The sulfur reacts with oxygen during combustion.
This forms sulfur dioxide.
Example 2 (Application):
Write a balanced equation for the formation of sulfur dioxide from sulfur.
▶️ Answer/Explanation
\( \mathrm{S + O_2 \rightarrow SO_2} \)
Example 3 (Hard):
Explain fully how burning a sulfur-containing fuel can lead to acid rain.
▶️ Answer/Explanation
Some fuels contain sulfur as an impurity.
During combustion, sulfur reacts with oxygen to form sulfur dioxide \( \mathrm{SO_2} \).
Sulfur dioxide is released into the atmosphere.
It can dissolve in rainwater to form acidic solutions.
This leads to acid rain.
Acid rain damages plants, buildings and aquatic life.