Edexcel iGCSE Chemistry -4.16 Acid Rain- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.16 Acid Rain- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.16 Acid Rain- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.16 understand how sulfur dioxide and oxides of nitrogen contribute to acid rain
4.16 How Sulfur Dioxide and Oxides of Nitrogen Contribute to Acid Rain
Acid rain is rainwater that has become more acidic than normal due to dissolved acidic gases.
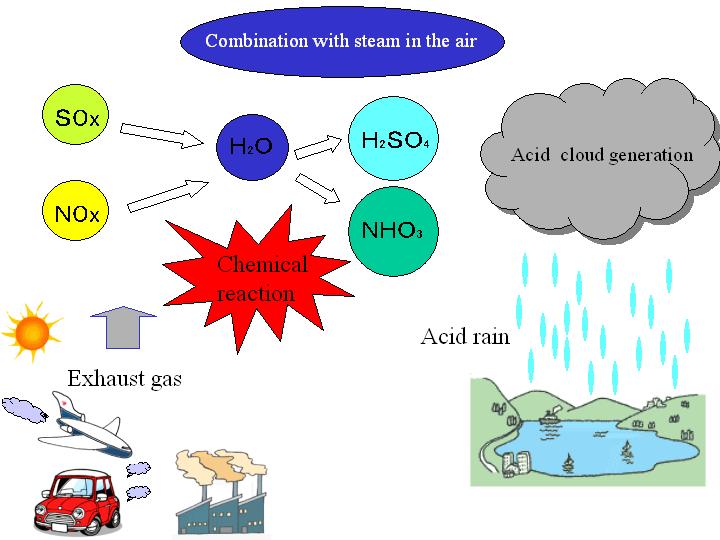
Two important gases that cause acid rain are:
- Sulfur dioxide \( \mathrm{SO_2} \)
- Oxides of nitrogen (e.g. \( \mathrm{NO} \), \( \mathrm{NO_2} \))
Step 1: Release of Acidic Gases
- \( \mathrm{SO_2} \) is formed when sulfur impurities in fuels burn.
- \( \mathrm{NO} \) and \( \mathrm{NO_2} \) form at high temperatures in car engines.
Step 2: Reaction with Water in the Atmosphere
Sulfur dioxide dissolves in water and reacts with oxygen to form sulfuric acid.
\( \mathrm{2SO_2 + O_2 \rightarrow 2SO_3} \)
\( \mathrm{SO_3 + H_2O \rightarrow H_2SO_4} \)
Oxides of nitrogen dissolve in water to form nitric acid.
\( \mathrm{4NO_2 + 2H_2O + O_2 \rightarrow 4HNO_3} \)
Step 3: Formation of Acid Rain
The acids formed dissolve in rainwater.
This lowers the pH of rainwater.
| Gas | Source | Acid Formed |
|---|---|---|
| \( \mathrm{SO_2} \) | Burning sulfur impurities in fuels | Sulfuric acid \( \mathrm{H_2SO_4} \) |
| \( \mathrm{NO_x} \) | High-temperature engine reactions | Nitric acid \( \mathrm{HNO_3} \) |
Effects of Acid Rain
- Damages trees and crops.
- Kills fish and aquatic organisms.
- Corrodes buildings made of limestone or marble.
- Acidifies soil.
Example 1 (Conceptual):
Why does acid rain have a lower pH than normal rain?
▶️ Answer/Explanation
Sulfur dioxide and nitrogen oxides dissolve in rainwater.
They form sulfuric acid and nitric acid.
This lowers the pH.
Example 2 (Application):
State one gas from car engines that contributes to acid rain.
▶️ Answer/Explanation
Nitrogen dioxide \( \mathrm{NO_2} \).
Example 3 (Hard):
Explain fully how sulfur dioxide from burning fuels leads to acid rain.
▶️ Answer/Explanation
Some fuels contain sulfur impurities.
When the fuel burns, sulfur reacts with oxygen to form sulfur dioxide \( \mathrm{SO_2} \).
This gas is released into the atmosphere.
It reacts with oxygen and water vapour to form sulfuric acid.
The acid dissolves in rainwater.
This lowers the pH of rain, forming acid rain.
