Edexcel iGCSE Chemistry -4.17–4.18 Catalytic Cracking and Its Importance- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.17–4.18 Catalytic Cracking and Its Importance- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.17–4.18 Catalytic Cracking and Its Importance- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.17 describe how long-chain alkanes are converted to alkenes and shorter-chain alkanes by catalytic cracking (using silica or alumina as the catalyst and a temperature in the range of 600–700 °C)
4.18 explain why cracking is necessary, in terms of the balance between supply and demand for different fractions
4.17 Catalytic Cracking of Long-Chain Alkanes
Catalytic cracking is the process used to break down long-chain alkanes into: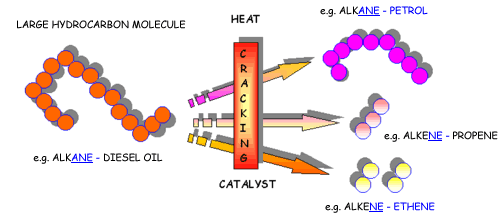
- Shorter-chain alkanes
- Alkenes
This process increases the supply of more useful and more valuable hydrocarbons.
Why Cracking Is Needed
- Long-chain hydrocarbons are less useful.
- They are less flammable.
- They have high boiling points and high viscosity.
- There is greater demand for shorter-chain fuels and alkenes.
Conditions for Catalytic Cracking
- Temperature: \( 600\text{–}700^\circ \mathrm{C} \)
- Catalyst: silica \( \mathrm{SiO_2} \) or alumina \( \mathrm{Al_2O_3} \)
- Vapour phase (hydrocarbon vapour)
What Happens During Cracking
Large alkane molecules are broken into smaller molecules.
Example:
\( \mathrm{C_{10}H_{22} \rightarrow C_8H_{18} + C_2H_4} \)
A long-chain alkane produces:
- A shorter alkane
- An alkene (contains a C=C double bond)
Role of the Catalyst
- Provides a surface for the reaction.
- Lowers the activation energy.
- Speeds up the reaction.
- Remains chemically unchanged.
| Before Cracking | After Cracking |
|---|---|
| Long-chain alkane | Shorter-chain alkane |
| High boiling point | Lower boiling point |
| Low demand | High demand products |
Why Alkenes Are Important
- Used to make polymers (plastics).
- Undergo addition reactions.
- More reactive than alkanes.
Exam Structure for Explanation
- Long-chain alkane is heated to \( 600\text{–}700^\circ \mathrm{C} \).
- Vapour passes over silica or alumina catalyst.
- Molecules break into smaller molecules.
- Products include alkanes and alkenes.
Example 1 (Conceptual):
Why is catalytic cracking used in industry?
▶️ Answer/Explanation
There is high demand for shorter-chain hydrocarbons and alkenes.
Cracking converts less useful long-chain alkanes into more valuable products.
Example 2 (Application):
One product of cracking must contain a double bond. Why?
▶️ Answer/Explanation
The original alkane is saturated.
When it splits, not all fragments can remain saturated.
At least one fragment forms an alkene with a C=C bond.
Example 3 (Hard):
Explain fully how long-chain alkanes are converted into alkenes and shorter-chain alkanes by catalytic cracking.
▶️ Answer/Explanation
Long-chain alkanes are first vaporised.
The vapour is heated to \( 600\text{–}700^\circ \mathrm{C} \).
It passes over a silica or alumina catalyst.
The catalyst lowers the activation energy.
The large molecules break into smaller molecules.
The products formed include shorter-chain alkanes and alkenes.
The alkenes contain a carbon–carbon double bond.
4.18 Why Cracking Is Necessary (Supply and Demand)
Crude oil contains a mixture of hydrocarbons that are separated into fractions by fractional distillation.
However, the amounts of each fraction produced do not match the demand for each fraction.
The Problem: Imbalance of Supply and Demand
- There is high demand for short-chain hydrocarbons (e.g. petrol).
- There is lower demand for long-chain hydrocarbons (e.g. fuel oil).
- Fractional distillation produces large amounts of long-chain fractions.
This creates a surplus of long-chain hydrocarbons and a shortage of short-chain hydrocarbons.
The Solution: Cracking
Cracking breaks long-chain alkanes into:
- Shorter-chain alkanes (useful fuels)
- Alkenes (used to make plastics)
Example:
\( \mathrm{C_{12}H_{26} \rightarrow C_8H_{18} + C_4H_8} \)
Economic Importance
- Increases the supply of high-demand fuels (e.g. gasoline).
- Produces alkenes for the polymer industry.
- Reduces waste of low-demand heavy fractions.
- Makes oil refining more profitable.
| Fraction Type | Supply | Demand |
|---|---|---|
| Short-chain (petrol) | Lower natural supply | High demand |
| Long-chain (fuel oil) | High natural supply | Lower demand |
Example 1 (Conceptual):
Why is there a need to convert long-chain hydrocarbons into shorter ones?
▶️ Answer/Explanation
There is higher demand for shorter-chain fuels.
There is excess supply of long-chain hydrocarbons.
Example 2 (Application):
Which fraction is in higher demand: petrol or fuel oil?
▶️ Answer/Explanation
Petrol.
It is widely used as fuel for cars.
Example 3 (Hard):
Explain fully why cracking is necessary in the oil industry.
▶️ Answer/Explanation
Fractional distillation produces more long-chain hydrocarbons than are needed.
There is greater demand for short-chain hydrocarbons such as petrol.
There is also demand for alkenes used to make plastics.
Cracking breaks long-chain hydrocarbons into shorter-chain alkanes and alkenes.
This increases the supply of high-demand products.
Therefore cracking balances supply and demand.
