Edexcel iGCSE Chemistry -4.19 General Formula of Alkanes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.19 General Formula of Alkanes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.19 General Formula of Alkanes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.19 know the general formula for alkanes
4.19 General Formula for Alkanes
Alkanes are a homologous series of hydrocarbons.
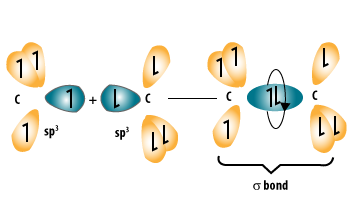
They contain:
- Only single carbon–carbon bonds.
- No double or triple bonds.
- Only carbon and hydrogen atoms.
General Formula
\( \mathrm{C_{n}H_{2n+2}} \)
Where:
- \( n \) = number of carbon atoms.
- \( 2n + 2 \) = number of hydrogen atoms.
Examples
If \( n = 1 \):
\( \mathrm{C_1H_{2(1)+2}} = \mathrm{CH_4} \) (methane)
If \( n = 2 \):
\( \mathrm{C_2H_{2(2)+2}} = \mathrm{C_2H_6} \) (ethane)
If \( n = 3 \):
\( \mathrm{C_3H_{2(3)+2}} = \mathrm{C_3H_8} \) (propane)
Why This Formula Works
Each carbon atom forms four covalent bonds.
In a saturated chain:
- Carbons bond to neighbouring carbons.
- Remaining bonds are filled by hydrogen.
This gives the pattern \( \mathrm{C_{n}H_{2n+2}} \).
| Number of Carbons (n) | Formula from \( \mathrm{C_{n}H_{2n+2}} \) | Name |
|---|---|---|
| 1 | \( \mathrm{CH_4} \) | Methane |
| 2 | \( \mathrm{C_2H_6} \) | Ethane |
| 4 | \( \mathrm{C_4H_{10}} \) | Butane |
Exam Key Points
- Applies only to alkanes (saturated hydrocarbons).
- If a formula does not match \( \mathrm{C_{n}H_{2n+2}} \), it is not an alkane.
- Alkanes contain only single bonds.
Example 1 (Conceptual):
Why is \( \mathrm{C_2H_4} \) not an alkane?
▶️ Answer/Explanation
For \( n = 2 \), the alkane formula gives \( \mathrm{C_2H_6} \).
\( \mathrm{C_2H_4} \) does not match \( \mathrm{C_{n}H_{2n+2}} \).
Therefore it is not an alkane.
Example 2 (Application):
Write the molecular formula of the alkane with 5 carbon atoms.
▶️ Answer/Explanation
Substitute \( n = 5 \):
\( \mathrm{C_5H_{2(5)+2}} = \mathrm{C_5H_{12}} \)
Name: pentane.
Example 3 (Hard):
A compound has the formula \( \mathrm{C_6H_{14}} \). Explain fully why this compound is an alkane.
▶️ Answer/Explanation
For \( n = 6 \), the general formula for alkanes gives:
\( \mathrm{C_6H_{2(6)+2}} = \mathrm{C_6H_{14}} \)
The formula matches \( \mathrm{C_{n}H_{2n+2}} \).
This means it is a saturated hydrocarbon.
Therefore it is an alkane.
