Edexcel iGCSE Chemistry -4.2 Representing Organic Molecules- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.2 Representing Organic Molecules- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.2 Representing Organic Molecules- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.2 understand how to represent organic molecules using empirical formulae, molecular formulae, general formulae, structural formulae and displayed formulae
4.2 Representing Organic Molecules
Organic molecules can be represented in several different ways. Each type of formula provides different information about the compound.
1. Empirical Formula
The empirical formula shows the simplest whole-number ratio of atoms in a compound.
Example:
Molecular formula of ethene: \( \mathrm{C_2H_4} \)
Empirical formula: \( \mathrm{CH_2} \)
The ratio \( \mathrm{C:H} = 2:4 \) simplifies to \( 1:2 \).
2. Molecular Formula
The molecular formula shows the actual number of atoms of each element in one molecule.
Ethane: \( \mathrm{C_2H_6} \)
Propane: \( \mathrm{C_3H_8} \)
It does not show how atoms are arranged.
3. General Formula
A general formula represents all members of a homologous series.
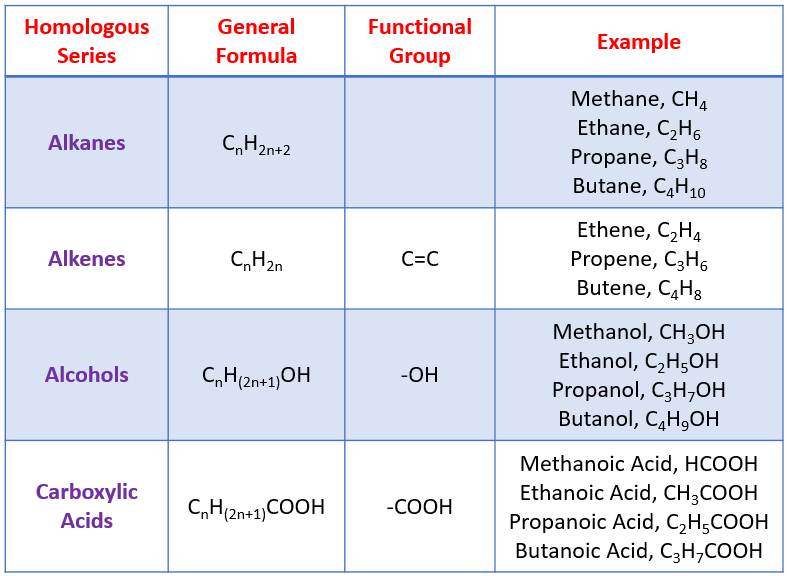
Example: Alkanes
\( \mathrm{C_{n}H_{2n+2}} \)
If \( n = 4 \):
\( \mathrm{C_4H_{10}} \)
4. Structural Formula
The structural formula shows how atoms are arranged in groups, but does not display every bond.
Example: Ethane
\( \mathrm{CH_3CH_3} \)
Example: Ethanol
\( \mathrm{CH_3CH_2OH} \)
5. Displayed Formula
The displayed formula shows all atoms and all bonds.
Example: Ethane
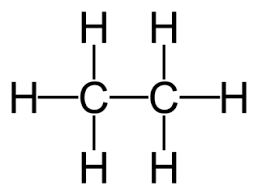
Every covalent bond is shown as a line.
| Type of Formula | What It Shows | Example (Ethene) |
|---|---|---|
| Empirical | Simplest ratio | \( \mathrm{CH_2} \) |
| Molecular | Actual numbers | \( \mathrm{C_2H_4} \) |
| General | Series pattern | \( \mathrm{C_{n}H_{2n}} \) |
| Structural | Atom grouping | \( \mathrm{CH_2CH_2} \) |
| Displayed | All bonds shown | H₂C=CH₂ (with bonds shown) |
Example 1 (Conceptual):
Why does the empirical formula not show the actual number of atoms?
▶️ Answer/Explanation
Because it shows only the simplest ratio of elements.
It does not give the exact number of atoms in a molecule.
Example 2 (Application):
State the empirical formula of \( \mathrm{C_3H_6} \).
▶️ Answer/Explanation
The ratio 3:6 simplifies to 1:2.
Empirical formula = \( \mathrm{CH_2} \).
Example 3 (Hard):
Explain fully the difference between the molecular formula and the displayed formula of \( \mathrm{C_2H_6} \).
▶️ Answer/Explanation
The molecular formula \( \mathrm{C_2H_6} \) shows that the molecule contains two carbon atoms and six hydrogen atoms.
It does not show how they are arranged.
The displayed formula shows every covalent bond between atoms.
It shows each carbon atom bonded to three hydrogen atoms and to each other.
Therefore the displayed formula gives structural information, while the molecular formula gives only numbers.
