Edexcel iGCSE Chemistry -4.20 Saturated Hydrocarbons- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.20 Saturated Hydrocarbons- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.20 Saturated Hydrocarbons- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.20 explain why alkanes are classified as saturated hydrocarbons
4.20 Why Alkanes Are Classified as Saturated Hydrocarbons
Alkanes are a homologous series of hydrocarbons with the general formula:
\( \mathrm{C_{n}H_{2n+2}} \)
Definition of Saturated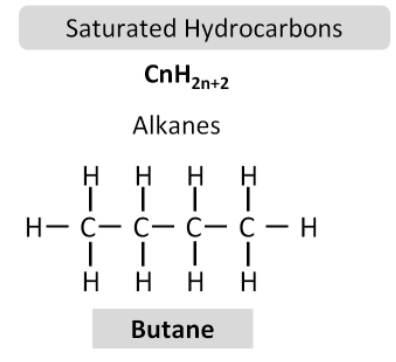
A saturated hydrocarbon is a compound that:
- Contains only single carbon–carbon bonds.
- Has the maximum number of hydrogen atoms attached to each carbon.
- Contains no double or triple bonds.
Why Alkanes Are Saturated
- Each carbon atom forms four covalent bonds.
- All carbon–carbon bonds are single bonds.
- The remaining bonds are filled with hydrogen atoms.
- No additional hydrogen atoms can be added without breaking bonds.
Therefore, alkanes are described as saturated.
Comparison: Saturated vs Unsaturated
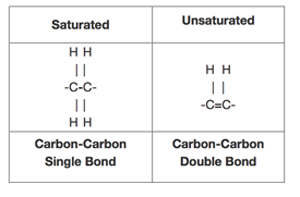
| Feature | Alkane | Alkene |
|---|---|---|
| Bond type | Single bonds only | Contains C=C |
| Hydrogen content | Maximum possible | Fewer hydrogens |
| General formula | \( \mathrm{C_{n}H_{2n+2}} \) | \( \mathrm{C_{n}H_{2n}} \) |
Example Structure
Ethane:
\( \mathrm{C_2H_6} \)
All carbon–carbon bonds are single bonds.
Example 1 (Conceptual):
Why is ethene not saturated?
▶️ Answer/Explanation
It contains a C=C double bond.
Therefore it does not have the maximum number of hydrogen atoms.
Example 2 (Application):
State one reason why \( \mathrm{C_3H_8} \) is classified as saturated.
▶️ Answer/Explanation
It contains only single carbon–carbon bonds.
Example 3 (Hard):
Explain fully why alkanes are described as saturated hydrocarbons.
▶️ Answer/Explanation
Alkanes are hydrocarbons containing only carbon and hydrogen.
They contain only single carbon–carbon bonds.
Each carbon forms four covalent bonds.
The remaining bonds are filled with hydrogen atoms.
This means they contain the maximum possible number of hydrogen atoms.
Therefore they are described as saturated.
