Edexcel iGCSE Chemistry -4.21 Structures and Naming of Alkanes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.21 Structures and Naming of Alkanes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.21 Structures and Naming of Alkanes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.21 understand how to draw the structural and displayed formulae for alkanes with up to five carbon atoms in the molecule, and to name the unbranched-chain isomers
4.21 Drawing Structural and Displayed Formulae of Alkanes (Up to Five Carbon Atoms) and Naming Unbranched-Chain Isomers
Alkanes are saturated hydrocarbons with general formula: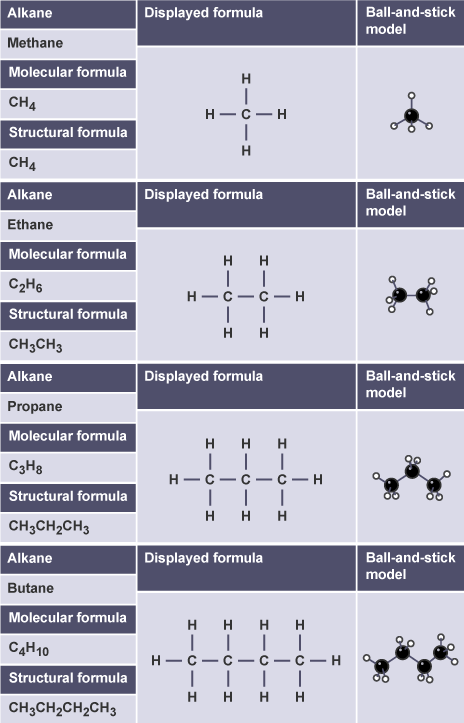
\( \mathrm{C_{n}H_{2n+2}} \)
You must be able to:
- Draw structural formulae.
- Draw displayed formulae.
- Name unbranched-chain isomers up to \( \mathrm{C_5} \).
1. Structural Formula
Shows the arrangement of atoms but not all bonds.
Example: Butane
\( \mathrm{CH_3CH_2CH_2CH_3} \)
2. Displayed Formula
Shows every atom and every covalent bond.
Example: Butane
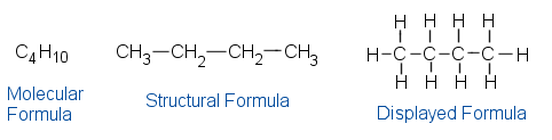
Names of Unbranched Alkanes (Up to 5 Carbons)
| Number of Carbons | Formula | Name |
|---|---|---|
| 1 | \( \mathrm{CH_4} \) | Methane |
| 2 | \( \mathrm{C_2H_6} \) | Ethane |
| 3 | \( \mathrm{C_3H_8} \) | Propane |
| 4 | \( \mathrm{C_4H_{10}} \) | Butane |
| 5 | \( \mathrm{C_5H_{12}} \) | Pentane |
Important Naming Rule
- Name depends on the number of carbon atoms in the longest chain.
- Unbranched means carbon atoms are in a single continuous chain.
- All alkanes end in -ane.
Exam Points
- Displayed formula shows all bonds.
- Structural formula shows condensed groups.
- Must count carbon atoms correctly before naming.
Example 1 (Conceptual):
Why is pentane classified as an alkane?
▶️ Answer/Explanation
It follows the general formula \( \mathrm{C_{n}H_{2n+2}} \).
It contains only single carbon–carbon bonds.
Example 2 (Application):
Draw the structural formula for propane.
▶️ Answer/Explanation
\( \mathrm{CH_3CH_2CH_3} \)
Example 3 (Hard):
A compound has the molecular formula \( \mathrm{C_4H_{10}} \). Draw its structural formula and name the unbranched-chain isomer.
▶️ Answer/Explanation
Using the alkane formula:
\( \mathrm{C_4H_{10}} \)
The unbranched chain structural formula is:
\( \mathrm{CH_3CH_2CH_2CH_3} \)
The name is butane.
