Edexcel iGCSE Chemistry -4.22 Reactions of Alkanes with Halogens- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.22 Reactions of Alkanes with Halogens- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.22 Reactions of Alkanes with Halogens- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.22 describe the reactions of alkanes with halogens in the presence of ultraviolet radiation, limited to mono-substitution
(knowledge of reaction mechanisms is not required)
4.22 Reaction of Alkanes with Halogens (Mono-substitution)
Alkanes undergo substitution reactions with halogens such as chlorine \( \mathrm{Cl_2} \) and bromine \( \mathrm{Br_2} \).
The reaction requires ultraviolet (UV) radiation.
What Type of Reaction?
It is called a substitution reaction because:
- One hydrogen atom in the alkane is replaced by a halogen atom.
- The rest of the molecule remains unchanged.
Only mono-substitution is required at GCSE level (one hydrogen replaced).
Example: Methane and Chlorine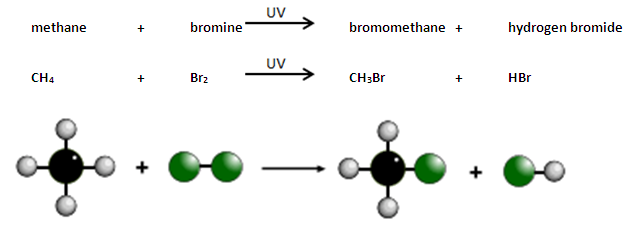
In UV light:
\( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
Products:
- Chloromethane \( \mathrm{CH_3Cl} \)
- Hydrogen chloride \( \mathrm{HCl} \)
Example: Ethane and Bromine
\( \mathrm{C_2H_6 + Br_2 \rightarrow C_2H_5Br + HBr} \)
Role of UV Radiation
- Provides energy to start the reaction.
- Breaks the bond in the halogen molecule.
- Without UV light, the reaction is very slow.
| Reactants | Condition | Products (Mono-substitution) |
|---|---|---|
| Alkane + Chlorine | UV light | Chloroalkane + \( \mathrm{HCl} \) |
| Alkane + Bromine | UV light | Bromoalkane + \( \mathrm{HBr} \) |
Exam Points
- Reaction type: substitution.
- Requires ultraviolet radiation.
- One hydrogen is replaced by a halogen.
- Produces a haloalkane and hydrogen halide.
Example 1 (Conceptual):
Why is the reaction between methane and chlorine called a substitution reaction?
▶️ Answer/Explanation
Because one hydrogen atom in methane is replaced by a chlorine atom.
The rest of the molecule remains the same.
Example 2 (Application):
Write the balanced equation for the mono-substitution reaction between ethane and chlorine.
▶️ Answer/Explanation
\( \mathrm{C_2H_6 + Cl_2 \rightarrow C_2H_5Cl + HCl} \)
Example 3 (Hard ):
Explain fully what happens when propane reacts with bromine in the presence of ultraviolet radiation.
▶️ Answer/Explanation
Propane is an alkane containing only single bonds.
In the presence of ultraviolet radiation, it reacts with bromine.
One hydrogen atom is replaced by a bromine atom.
This is a substitution reaction.
A bromoalkane and hydrogen bromide \( \mathrm{HBr} \) are formed.
The reaction requires UV light to begin.
