Edexcel iGCSE Chemistry -4.23–4.24 Functional Group & General Formula of Alkenes- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.23–4.24 Functional Group & General Formula of Alkenes- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.23–4.24 Functional Group & General Formula of Alkenes- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.23 know that alkenes contain the functional group >C=C<
4.24 know the general formula for alkenes
4.23 Alkenes and the Functional Group >C=C<
Alkenes are a homologous series of hydrocarbons.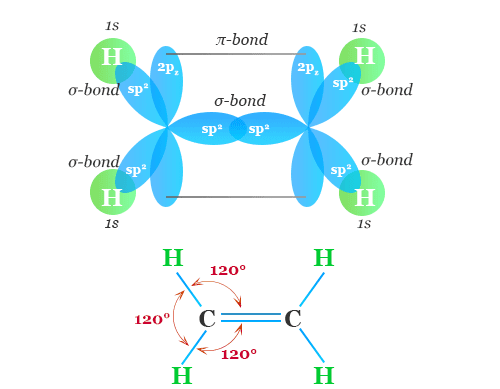
They contain the functional group:
>C=C<
What Is a Functional Group?
A functional group is a group of atoms responsible for the characteristic chemical reactions of a compound.
For alkenes, the functional group is the carbon–carbon double bond.
Key Features of Alkenes
- Contain at least one C=C double bond.
- Are unsaturated hydrocarbons.
- Have general formula \( \mathrm{C_{n}H_{2n}} \) (for one double bond).
- Undergo addition reactions.
Examples
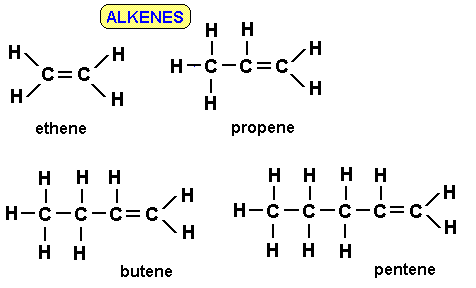
Ethene:
\( \mathrm{C_2H_4} \)
Structural formula:
\( \mathrm{CH_2=CH_2} \)
Propene:
\( \mathrm{C_3H_6} \)
Why the Double Bond Is Important
- It makes alkenes more reactive than alkanes.
- It allows addition reactions to occur.
- It means alkenes are unsaturated.
| Feature | Alkane | Alkene |
|---|---|---|
| Functional group | None | >C=C< |
| Bond type | Single bonds only | At least one double bond |
| General formula | \( \mathrm{C_{n}H_{2n+2}} \) | \( \mathrm{C_{n}H_{2n}} \) |
Key Points
- Alkenes contain a C=C double bond.
- This is the functional group.
- It determines their chemical behaviour.
Example 1 (Conceptual):
Why is ethene classified as an alkene?
▶️ Answer/Explanation
It contains a carbon–carbon double bond.
This is the functional group of alkenes.
Example 2 (Application):
State the functional group present in propene.
▶️ Answer/Explanation
The carbon–carbon double bond (>C=C<).
Example 3 (Hard):
Explain fully why alkenes are described as unsaturated hydrocarbons.
▶️ Answer/Explanation
Alkenes are hydrocarbons containing only carbon and hydrogen.
They contain at least one carbon–carbon double bond.
This means they do not have the maximum possible number of hydrogen atoms.
The double bond is the functional group.
Therefore they are unsaturated hydrocarbons.
4.24 General Formula for Alkenes
Alkenes are unsaturated hydrocarbons that contain the functional group:
>C=C<
General Formula
\( \mathrm{C_nH_{2n}} \)
This formula applies to alkenes containing one carbon–carbon double bond.
What the Formula Means
- \( n \) = number of carbon atoms.
- The number of hydrogen atoms is \( 2n \).
- There are two fewer hydrogen atoms than the corresponding alkane.
Why There Are Fewer Hydrogens
The presence of a carbon–carbon double bond means:
- Carbons share two pairs of electrons.
- Fewer hydrogen atoms are needed to complete four bonds per carbon.
- Therefore the molecule is unsaturated.
Examples
If \( n = 2 \):
\( \mathrm{C_2H_{2(2)}} = \mathrm{C_2H_4} \) (ethene)
If \( n = 3 \):
\( \mathrm{C_3H_{2(3)}} = \mathrm{C_3H_6} \) (propene)
If \( n = 4 \):
\( \mathrm{C_4H_8} \) (butene)
| Number of Carbons (n) | Alkane Formula | Alkene Formula |
|---|---|---|
| 2 | \( \mathrm{C_2H_6} \) | \( \mathrm{C_2H_4} \) |
| 3 | \( \mathrm{C_3H_8} \) | \( \mathrm{C_3H_6} \) |
| 4 | \( \mathrm{C_4H_{10}} \) | \( \mathrm{C_4H_8} \) |
Exam Key Points
- General formula for alkenes: \( \mathrm{C_nH_{2n}} \).
- Applies to one double bond.
- They contain two fewer hydrogen atoms than the corresponding alkane.
- Alkenes are unsaturated hydrocarbons.
Example 1 (Conceptual):
Why does \( \mathrm{C_3H_6} \) fit the general formula of an alkene?
▶️ Answer/Explanation
For \( n = 3 \), the formula gives \( \mathrm{C_3H_{2(3)}} = \mathrm{C_3H_6} \).
Therefore it matches \( \mathrm{C_nH_{2n}} \).
Example 2 (Application):
Write the molecular formula of an alkene with 5 carbon atoms.
▶️ Answer/Explanation
Substitute \( n = 5 \):
\( \mathrm{C_5H_{2(5)}} = \mathrm{C_5H_{10}} \)
Example 3 (Hard):
A compound has the formula \( \mathrm{C_4H_8} \). Explain fully why it could be an alkene but \( \mathrm{C_4H_{10}} \) cannot.
▶️ Answer/Explanation
For \( n = 4 \), the alkene formula gives \( \mathrm{C_4H_{2(4)}} = \mathrm{C_4H_8} \).
This matches the general formula \( \mathrm{C_nH_{2n}} \).
This suggests the molecule may contain a carbon–carbon double bond.
However, \( \mathrm{C_4H_{10}} \) fits the alkane formula \( \mathrm{C_nH_{2n+2}} \).
Therefore \( \mathrm{C_4H_{10}} \) is an alkane, not an alkene.
