Edexcel iGCSE Chemistry -4.25 Unsaturated Hydrocarbons- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.25 Unsaturated Hydrocarbons- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.25 Unsaturated Hydrocarbons- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.25 explain why alkenes are classified as unsaturated hydrocarbons
4.25 Why Alkenes Are Classified as Unsaturated Hydrocarbons
Alkenes are a homologous series of hydrocarbons that contain the functional group:
>C=C<
Definition of Unsaturated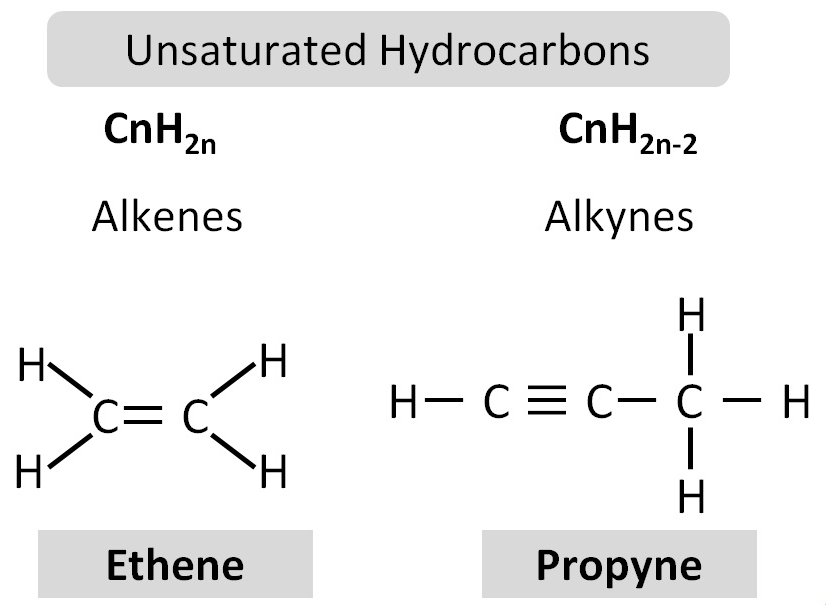
An unsaturated hydrocarbon is a compound that:
- Contains at least one carbon–carbon double bond.
- Does not contain the maximum possible number of hydrogen atoms.
Why Alkenes Are Unsaturated
- They contain a C=C double bond.
- Each carbon in the double bond shares two pairs of electrons.
- This means fewer hydrogen atoms are bonded to the carbon atoms.
- Therefore, more atoms could be added across the double bond.
Because they can undergo addition reactions, they are not fully saturated with hydrogen.
Comparison: Saturated vs Unsaturated
| Feature | Alkane (Saturated) | Alkene (Unsaturated) |
|---|---|---|
| Bond type | Single bonds only | Contains C=C double bond |
| Hydrogen content | Maximum possible | Fewer hydrogen atoms |
| Reactivity | Less reactive | More reactive |
Example Structure
Ethene:
\( \mathrm{CH_2 = CH_2} \)
The double bond shows it is unsaturated.
Keywords to Include
- Contains a carbon–carbon double bond.
- Does not have maximum number of hydrogen atoms.
- Can undergo addition reactions.
Example 1 (Conceptual):
Why is ethene described as unsaturated?
▶️ Answer/Explanation
It contains a carbon–carbon double bond.
It does not contain the maximum possible number of hydrogen atoms.
Example 2 (Application):
Which molecule is unsaturated: \( \mathrm{C_2H_6} \) or \( \mathrm{C_2H_4} \)?
▶️ Answer/Explanation
\( \mathrm{C_2H_4} \) is unsaturated.
It fits the alkene formula \( \mathrm{C_{n}H_{2n}} \).
It contains a double bond.
Example 3 (Hard ):
Explain fully why alkenes are classified as unsaturated hydrocarbons.
▶️ Answer/Explanation
Alkenes are hydrocarbons made of carbon and hydrogen.
They contain at least one carbon–carbon double bond.
Because of the double bond, they do not have the maximum possible number of hydrogen atoms.
The double bond allows addition reactions to occur.
Therefore they are described as unsaturated hydrocarbons.
