Edexcel iGCSE Chemistry -4.27–4.28 Reactions with Bromine and the Bromine Water Test- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.27–4.28 Reactions with Bromine and the Bromine Water Test- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.27–4.28 Reactions with Bromine and the Bromine Water Test- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.27 describe the reactions of alkenes with bromine to produce dibromoalkanes
4.28 describe how bromine water can be used to distinguish between an alkane and an alkene
4.27 Reaction of Alkenes with Bromine (Formation of Dibromoalkanes)
Alkenes undergo addition reactions because they contain a carbon–carbon double bond.
When an alkene reacts with bromine \( \mathrm{Br_2} \), a dibromoalkane is formed.
Type of Reaction
This is an addition reaction because: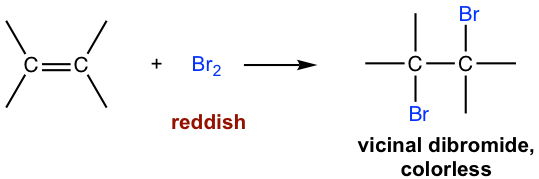
- The double bond breaks.
- Each carbon forms a new bond with a bromine atom.
- No other molecule is formed.
Example: Ethene and Bromine
\( \mathrm{C_2H_4 + Br_2 \rightarrow C_2H_4Br_2} \)
Product: 1,2-dibromoethane
What Happens to the Double Bond?
- The C=C double bond opens.
- Each carbon atom forms a single bond with bromine.
- The molecule becomes saturated.
Observation (Bromine Water Test)
- Bromine water is orange.
- When added to an alkene, it turns colourless.
- This confirms the presence of a C=C double bond.
Alkanes do not decolourise bromine water under normal conditions.
| Hydrocarbon | Reaction with Bromine Water | Result |
|---|---|---|
| Alkene | Addition reaction | Orange → colourless |
| Alkane | No reaction (without UV) | Remains orange |
Exam Key Points
- Reaction type: addition.
- Double bond breaks.
- Dibromoalkane forms.
- Bromine water test turns from orange to colourless.
Example 1 (Conceptual):
Why does bromine water turn colourless when added to an alkene?
▶️ Answer/Explanation
The alkene undergoes an addition reaction.
The C=C double bond breaks.
Bromine molecules are used up, so the orange colour disappears.
Example 2 (Application):
Write the balanced equation for the reaction between propene and bromine.
▶️ Answer/Explanation
\( \mathrm{C_3H_6 + Br_2 \rightarrow C_3H_6Br_2} \)
Product: dibromopropane.
Example 3 (Hard):
Explain fully how bromine water can be used to distinguish between an alkane and an alkene.
▶️ Answer/Explanation
Bromine water is orange in colour.
An alkene contains a carbon–carbon double bond.
The alkene reacts with bromine in an addition reaction.
The double bond breaks and a dibromoalkane is formed.
The bromine is used up, so the solution becomes colourless.
An alkane does not react without ultraviolet light.
Therefore the solution remains orange with an alkane.
4.28 Using Bromine Water to Distinguish Between an Alkane and an Alkene
Bromine water is an orange solution containing bromine \( \mathrm{Br_2} \).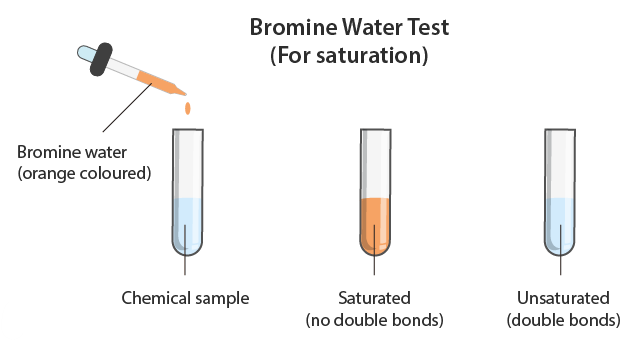
It is used as a test for unsaturation.
What Happens with an Alkene?
- Alkenes contain a carbon–carbon double bond.
- They undergo an addition reaction with bromine.
- The double bond breaks.
- A dibromoalkane is formed.
- The orange colour disappears.
Example (ethene): \( \mathrm{C_2H_4 + Br_2 \rightarrow C_2H_4Br_2} \)
Observation: Orange → colourless.
What Happens with an Alkane?
- Alkanes contain only single bonds.
- No double bond is present.
- No reaction occurs under normal conditions.
- The orange colour remains.
(Alkanes only react with bromine in UV light by substitution.)
| Hydrocarbon | Contains C=C? | Colour Change |
|---|---|---|
| Alkene | Yes | Orange → colourless |
| Alkane | No | Remains orange |
Why the Colour Changes
- The alkene reacts with bromine.
- Bromine molecules are used up.
- The orange colour disappears.
Example 1 (Conceptual):
Why does an alkene decolourise bromine water?
▶️ Answer/Explanation
It contains a carbon–carbon double bond.
The double bond reacts with bromine in an addition reaction.
Bromine is used up, so the solution becomes colourless.
Example 2 (Application):
A hydrocarbon does not change the colour of bromine water. What type of hydrocarbon is it?
▶️ Answer/Explanation
It is an alkane.
Alkanes contain only single bonds and do not react without UV light.
Example 3 (Hard):
Explain fully how bromine water can distinguish between an alkane and an alkene.
▶️ Answer/Explanation
Bromine water is orange in colour.
An alkene contains a carbon–carbon double bond.
It reacts with bromine in an addition reaction.
The double bond breaks and a dibromoalkane is formed.
Bromine is used up so the solution turns colourless.
An alkane contains only single bonds.
It does not react under normal conditions, so the solution remains orange.
