Edexcel iGCSE Chemistry -4.29C Functional Group of Alcohols- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.29C Functional Group of Alcohols- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.29C Functional Group of Alcohols- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.29C know that alcohols contain the functional group −OH
4.29C Alcohols and the Functional Group −OH
Alcohols are a homologous series of organic compounds.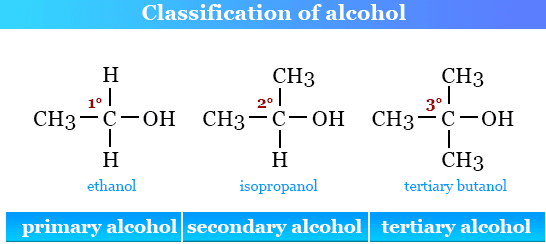
They contain the functional group:
−OH
This is called the hydroxyl group.
What Is a Functional Group?
A functional group is a group of atoms responsible for the characteristic chemical properties of a compound.
For alcohols, the −OH group determines their chemical behaviour.
General Formula of Alcohols
For alcohols based on alkanes:
\( \mathrm{C_{n}H_{2n+1}OH} \)
Or sometimes written as:
\( \mathrm{C_{n}H_{2n+2}O} \)
Examples
Methanol
\( \mathrm{CH_3OH} \)
Ethanol
\( \mathrm{C_2H_5OH} \)
Propanol
\( \mathrm{C_3H_7OH} \)
Important Features of Alcohols
- Contain carbon, hydrogen and oxygen.
- Have at least one −OH group.
- Names end in -ol.
- Are different from alkanes because they contain oxygen.
| Compound | Contains −OH? | Class |
|---|---|---|
| \( \mathrm{C_2H_6} \) | No | Alkane |
| \( \mathrm{C_2H_5OH} \) | Yes | Alcohol |
Key Points
- Functional group of alcohols is −OH.
- This is called the hydroxyl group.
- Alcohol names end in -ol.
Example 1 (Conceptual):
Why is ethanol classified as an alcohol?
▶️ Answer/Explanation
It contains the −OH functional group.
The −OH group defines alcohols.
Example 2 (Application):
Identify the functional group present in methanol.
▶️ Answer/Explanation
The hydroxyl group (−OH).
Example 3 (Hard ):
Explain fully how alcohols differ from alkanes.
▶️ Answer/Explanation
Alkanes contain only carbon and hydrogen and have only single bonds.
Alcohols contain carbon, hydrogen and oxygen.
Alcohols contain the −OH functional group.
This functional group gives alcohols different chemical properties.
Therefore alcohols are a different homologous series from alkanes.
