Edexcel iGCSE Chemistry -4.3 Homologous Series, Functional Groups and Isomerism- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.3 Homologous Series, Functional Groups and Isomerism- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.3 Homologous Series, Functional Groups and Isomerism- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.3 know what is meant by the terms homologous series, functional group and isomerism
4.3 Homologous Series, Functional Group and Isomerism
Homologous Series
A homologous series is a family of organic compounds that:
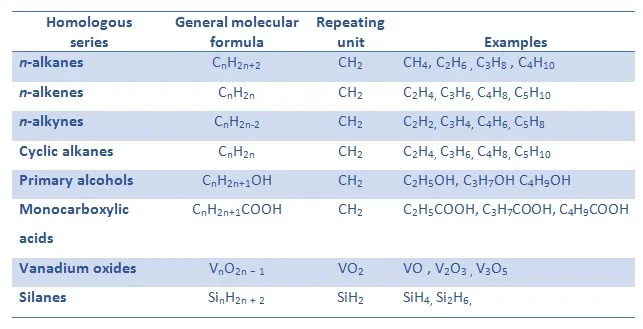
- Have the same general formula
- Have the same functional group
- Have similar chemical properties
- Show a gradual trend in physical properties (e.g. boiling point)
- Differ by \( \mathrm{CH_2} \) between successive members
Example: Alkanes
General formula: \( \mathrm{C_{n}H_{2n+2}} \)
Methane → \( \mathrm{CH_4} \) Ethane → \( \mathrm{C_2H_6} \) Propane → \( \mathrm{C_3H_8} \)
Each member increases by \( \mathrm{CH_2} \).
Functional Group
A functional group is a specific group of atoms within a molecule that determines its chemical reactions.
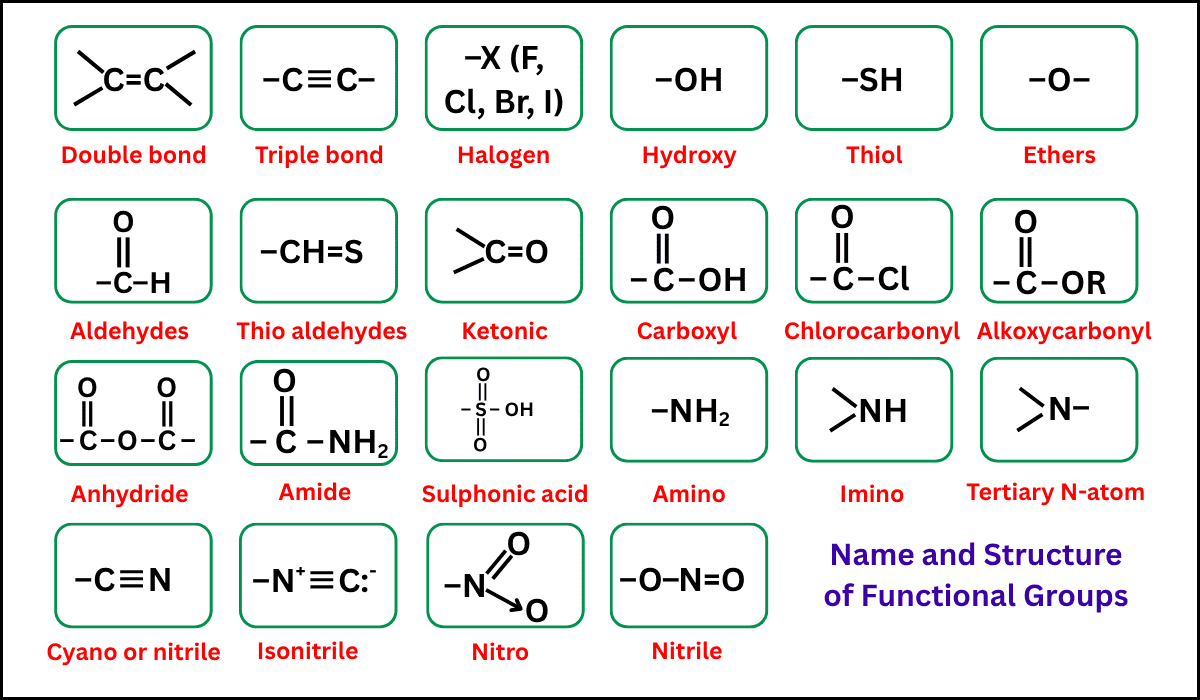
It gives compounds in the same homologous series similar chemical properties.
Examples:
| Homologous Series | Functional Group | Example |
|---|---|---|
| Alkanes | None (single C–C bonds only) | \( \mathrm{C_2H_6} \) |
| Alkenes | C=C double bond | \( \mathrm{C_2H_4} \) |
| Alcohols | \( \mathrm{-OH} \) | \( \mathrm{C_2H_5OH} \) |
Isomerism
Isomerism occurs when compounds have:

- The same molecular formula
- But different structural arrangements of atoms
These compounds are called isomers.
Example: \( \mathrm{C_4H_{10}} \)
Straight-chain butane and branched 2-methylpropane are structural isomers.
They have different physical properties (e.g. boiling points).
| Term | Definition |
|---|---|
| Homologous series | Family of compounds with same general formula and functional group |
| Functional group | Group of atoms responsible for reactions |
| Isomerism | Same molecular formula, different structure |
Example 1 (Conceptual):
Why do members of the same homologous series have similar chemical properties?
▶️ Answer/Explanation
Because they have the same functional group.
The functional group determines chemical reactions.
Example 2 (Application):
Which two statements describe isomers?
A. Same molecular formula B. Same structural formula C. Different arrangement of atoms D. Different molecular formula
▶️ Answer/Explanation
Correct answers: A and C.
Isomers have the same molecular formula but different arrangements.
Example 3 (Hard):
Explain fully how the term homologous series differs from isomerism.
▶️ Answer/Explanation
A homologous series is a family of compounds with the same general formula and functional group.
Each member differs by \( \mathrm{CH_2} \).
They have similar chemical properties.
Isomerism occurs within compounds that have the same molecular formula but different structures.
Isomers are different compounds but share the same molecular formula.
Therefore homologous series describe families of compounds, while isomerism describes structural differences within the same molecular formula.
