Edexcel iGCSE Chemistry -4.30C Structures and Naming of Alcohols- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.30C Structures and Naming of Alcohols- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.30C Structures and Naming of Alcohols- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.30C understand how to draw structural and displayed formulae for methanol, ethanol, propanol (propan-1-ol only) and butanol (butan-1-ol only), and name each compound
(the names propanol and butanol are acceptable)
4.30C Drawing and Naming Methanol, Ethanol, Propan-1-ol and Butan-1-ol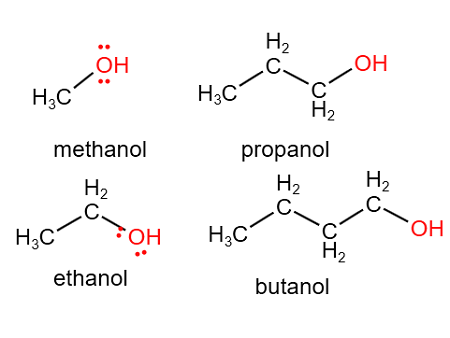
Alcohols contain the functional group:
−OH
For this specification, you must be able to:
- Draw structural formulae.
- Draw displayed formulae.
- Name methanol, ethanol, propan-1-ol and butan-1-ol.
- The names propanol and butanol are acceptable.
1. Methanol
Molecular formula:
\( \mathrm{CH_3OH} \)
Structural formula:
\( \mathrm{CH_3OH} \)
Displayed formula:
\( \mathrm{H} \)
\( \mathrm{\ |} \)
\( \mathrm{H – C – O – H} \)
\( \mathrm{\ |} \)
\( \mathrm{H} \)
2. Ethanol
Molecular formula:
\( \mathrm{C_2H_5OH} \)
Structural formula:
\( \mathrm{CH_3CH_2OH} \)
Displayed formula (simplified layout):
\( \mathrm{CH_3 – CH_2 – OH} \)
3. Propan-1-ol (Propanol)
Molecular formula:
\( \mathrm{C_3H_7OH} \)
Structural formula:
\( \mathrm{CH_3CH_2CH_2OH} \)
The −OH group is on carbon 1.
4. Butan-1-ol (Butanol)
Molecular formula:
\( \mathrm{C_4H_9OH} \)
Structural formula:
\( \mathrm{CH_3CH_2CH_2CH_2OH} \)
Again, the −OH group is on carbon 1.
Naming Rules
- Longest carbon chain determines root name.
- Replace “-ane” with “-ol”.
- Number carbon atoms so −OH has lowest possible number.
- If −OH is on carbon 1 → “-1-ol”.
| Compound | Structural Formula | Name |
|---|---|---|
| \( \mathrm{CH_3OH} \) | \( \mathrm{CH_3OH} \) | Methanol |
| \( \mathrm{C_2H_5OH} \) | \( \mathrm{CH_3CH_2OH} \) | Ethanol |
| \( \mathrm{C_3H_7OH} \) | \( \mathrm{CH_3CH_2CH_2OH} \) | Propan-1-ol |
| \( \mathrm{C_4H_9OH} \) | \( \mathrm{CH_3CH_2CH_2CH_2OH} \) | Butan-1-ol |
Key Points
- All contain −OH.
- Names end in “-ol”.
- −OH must be on carbon 1 for this specification.
- Displayed formula shows all bonds clearly.
Example 1 (Conceptual):
Why is propan-1-ol not classified as an alkane?
▶️ Answer/Explanation
It contains the −OH functional group.
Alkanes contain only carbon and hydrogen.
Example 2 (Application):
Write the structural formula for butan-1-ol.
▶️ Answer/Explanation
\( \mathrm{CH_3CH_2CH_2CH_2OH} \)
Example 3 (Hard ):
Explain fully how you would name \( \mathrm{CH_3CH_2CH_2OH} \).
▶️ Answer/Explanation
There are three carbon atoms, so the root name is propan-.
The molecule contains the −OH functional group.
The −OH group is on carbon 1.
The suffix for alcohols is -ol.
Therefore the name is propan-1-ol (propanol).
