Edexcel iGCSE Chemistry -4.31C Oxidation of Ethanol- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.31C Oxidation of Ethanol- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.31C Oxidation of Ethanol- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.31C know that ethanol can be oxidised by:
• burning in air or oxygen (complete combustion)
• reaction with oxygen in the air to form ethanoic acid (microbial oxidation)
• heating with potassium dichromate(VI) in dilute sulfuric acid to form ethanoic acid
4.31C Oxidation of Ethanol
Oxidation is defined as:
- Gain of oxygen
- Loss of electrons
Ethanol \( \mathrm{C_2H_5OH} \) can undergo oxidation in several different ways.
1. Complete Combustion (Burning in Air or Oxygen)
When ethanol burns in excess oxygen, it undergoes complete combustion.
\( \mathrm{C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O} \)
- Ethanol gains oxygen.
- Carbon dioxide and water are formed.
- Heat energy is released (exothermic).
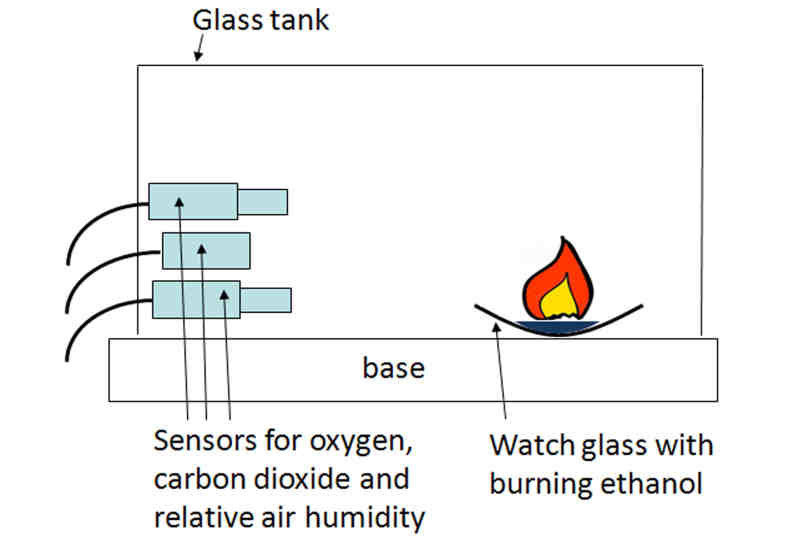
2. Microbial Oxidation (Reaction with Oxygen in Air)
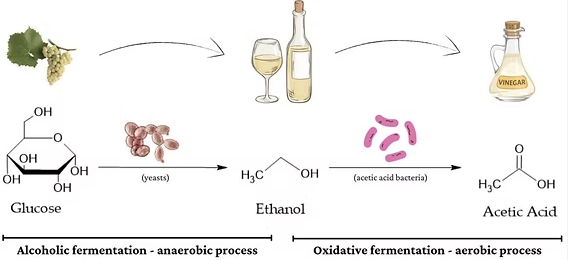
Ethanol can react slowly with oxygen in the air.
This occurs in the presence of bacteria (e.g. in vinegar production).
\( \mathrm{C_2H_5OH + O_2 \rightarrow CH_3COOH + H_2O} \)
- Ethanoic acid is formed.
- This process is called microbial oxidation.
3. Oxidation Using Potassium Dichromate(VI)
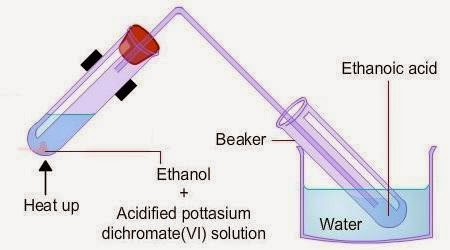
When ethanol is heated with potassium dichromate(VI) in dilute sulfuric acid:
\( \mathrm{C_2H_5OH + 2[O] \rightarrow CH_3COOH + H_2O} \)
- Ethanol is oxidised to ethanoic acid.
- The orange dichromate solution turns green.
- This colour change is evidence of oxidation.
| Method | Conditions | Product |
|---|---|---|
| Combustion | Burn in air/O₂ | \( \mathrm{CO_2 + H_2O} \) |
| Microbial oxidation | Oxygen in air + bacteria | \( \mathrm{CH_3COOH} \) |
| Chemical oxidation | \( \mathrm{K_2Cr_2O_7/H_2SO_4} \), heat | \( \mathrm{CH_3COOH} \) |
Exam Key Points
- Ethanol can be oxidised in three different ways.
- Combustion forms carbon dioxide and water.
- Slow oxidation in air forms ethanoic acid.
- Dichromate changes from orange to green.
Example 1 (Conceptual):
Why is burning ethanol considered oxidation?
▶️ Answer/Explanation
Ethanol gains oxygen.
Oxidation is defined as gain of oxygen.
Example 2 (Application):
State the colour change observed when ethanol is heated with potassium dichromate(VI).
▶️ Answer/Explanation
The solution changes from orange to green.
Example 3 (Hard):
Explain fully how ethanol can be converted into ethanoic acid.
▶️ Answer/Explanation
Ethanol can react slowly with oxygen in the air.
This is microbial oxidation.
Ethanoic acid is produced.
Ethanol can also be heated with potassium dichromate(VI) in dilute sulfuric acid.
It gains oxygen and is oxidised to ethanoic acid.
The dichromate changes from orange to green.
