Edexcel iGCSE Chemistry -4.32C Manufacture of Ethanol- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.32C Manufacture of Ethanol- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.32C Manufacture of Ethanol- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.32C know that ethanol can be manufactured by:
• reacting ethene with steam in the presence of a phosphoric acid catalyst at about 300 °C and 60–70 atm
• the fermentation of glucose, in the absence of air, at an optimum temperature of about 30 °C and using the enzymes in yeast
4.32C Manufacture of Ethanol
Ethanol \( \mathrm{C_2H_5OH} \) can be manufactured by two main methods:
- Hydration of ethene (industrial method)
- Fermentation of glucose (biological method)
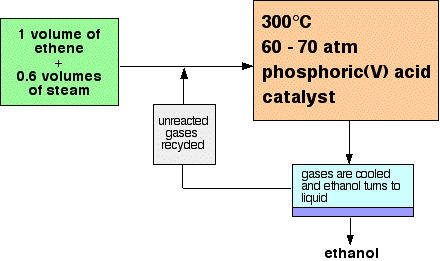
1. Hydration of Ethene (Industrial Method)
Ethene reacts with steam in the presence of a phosphoric acid catalyst.
\( \mathrm{C_2H_4 + H_2O \rightarrow C_2H_5OH} \)
Conditions:
- Temperature: about \( 300^\circ \mathrm{C} \)
- Pressure: about 60–70 atm
- Catalyst: phosphoric acid \( \mathrm{H_3PO_4} \)
This is an addition reaction because the double bond in ethene opens and water adds across it.
2. Fermentation of Glucose
Glucose is converted into ethanol and carbon dioxide by enzymes in yeast.
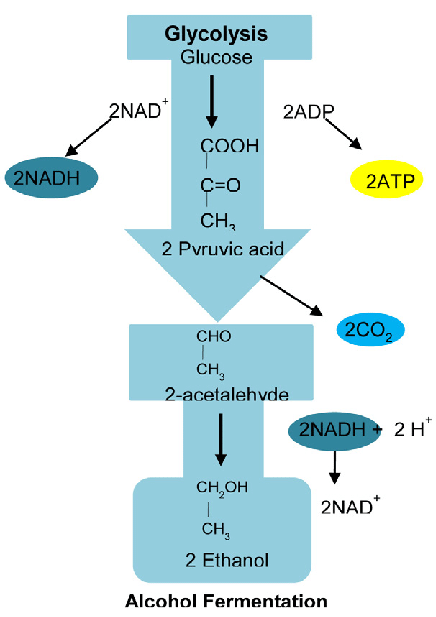
\( \mathrm{C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2CO_2} \)
Conditions:
- No oxygen (anaerobic conditions)
- Optimum temperature about \( 30^\circ \mathrm{C} \)
- Yeast enzymes as catalyst
If oxygen is present, the ethanol may be oxidised to ethanoic acid.
Comparison of the Two Methods
| Feature | Hydration of Ethene | Fermentation |
|---|---|---|
| Raw material | Ethene (from crude oil) | Glucose (from plants) |
| Temperature | \( 300^\circ \mathrm{C} \) | \( 30^\circ \mathrm{C} \) |
| Pressure | 60–70 atm | Atmospheric pressure |
| Catalyst | Phosphoric acid | Yeast enzymes |
| Speed | Fast | Slow |
Key Points
- Ethanol can be made by hydration of ethene.
- Requires high temperature and pressure.
- Ethanol can also be made by fermentation of glucose.
- Fermentation requires yeast and no oxygen.
Example 1 (Conceptual):
Why must fermentation occur in the absence of oxygen?
▶️ Answer/Explanation
If oxygen is present, ethanol can be oxidised further.
Anaerobic conditions are needed to produce ethanol.
Example 2 (Application):
Write the balanced equation for the fermentation of glucose.
▶️ Answer/Explanation
\( \mathrm{C_6H_{12}O_6 \rightarrow 2C_2H_5OH + 2CO_2} \)
Example 3 (Hard):
Explain fully how ethanol is manufactured by hydration of ethene.
▶️ Answer/Explanation
Ethene reacts with steam.
The reaction requires a phosphoric acid catalyst.
The temperature is about \( 300^\circ \mathrm{C} \).
The pressure is about 60–70 atm.
The double bond in ethene opens.
Water adds across the double bond.
Ethanol is formed.
