Edexcel iGCSE Chemistry -4.34C Functional Group of Carboxylic Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.34C Functional Group of Carboxylic Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.34C Functional Group of Carboxylic Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.34C know that carboxylic acids contain the functional group –COOH
4.34C Carboxylic Acids and Their Functional Group
Carboxylic acids are a homologous series of organic compounds.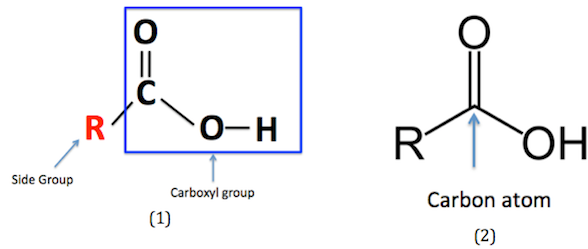
They contain the functional group:
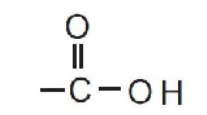
This is called the carboxyl group.
Structure of the Carboxyl Group
The −COOH group contains:
- A carbon atom.
- A carbon–oxygen double bond (C=O).
- A hydroxyl group (−OH).
So the structure can be represented as:
\( \mathrm{-C(=O)OH} \)
General Formula of Carboxylic Acids
\( \mathrm{C_{n}H_{2n+1}COOH} \)
Carboxylic acids contain carbon, hydrogen and oxygen.
Examples
Methanoic acid
\( \mathrm{HCOOH} \)
Ethanoic acid
\( \mathrm{CH_3COOH} \)
Ethanoic acid is the main acid in vinegar.
| Compound | Functional Group | Class |
|---|---|---|
| \( \mathrm{C_2H_5OH} \) | −OH | Alcohol |
| \( \mathrm{CH_3COOH} \) | −COOH | Carboxylic acid |
Key Properties of Carboxylic Acids
- Contain the −COOH functional group.
- Are weak acids.
- Can neutralise alkalis to form salts.
- Names end in -oic acid.
Key Points
- Functional group of carboxylic acids is −COOH.
- This group contains both C=O and −OH.
- Names end in “-oic acid”.
Example 1 (Conceptual):
Why is \( \mathrm{CH_3COOH} \) classified as a carboxylic acid?
▶️ Answer/Explanation
It contains the −COOH functional group.
The −COOH group defines carboxylic acids.
Example 2 (Application):
State the functional group present in methanoic acid.
▶️ Answer/Explanation
The carboxyl group (−COOH).
Example 3 (Hard ):
Explain fully why carboxylic acids are different from alcohols.
▶️ Answer/Explanation
Carboxylic acids contain the −COOH functional group.
This group contains a carbon–oxygen double bond and a hydroxyl group.
Alcohols contain only the −OH functional group.
Because of the −COOH group, carboxylic acids are acidic.
Alcohols are not acidic in the same way.
