Edexcel iGCSE Chemistry -4.35C Structures and Naming of Carboxylic Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.35C Structures and Naming of Carboxylic Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.35C Structures and Naming of Carboxylic Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.35C understand how to draw structural and displayed formulae for unbranched-chain carboxylic acids with up to four carbon atoms in the molecule, and name each compound
4.35C Drawing and Naming Unbranched-Chain Carboxylic Acids (Up to 4 Carbons)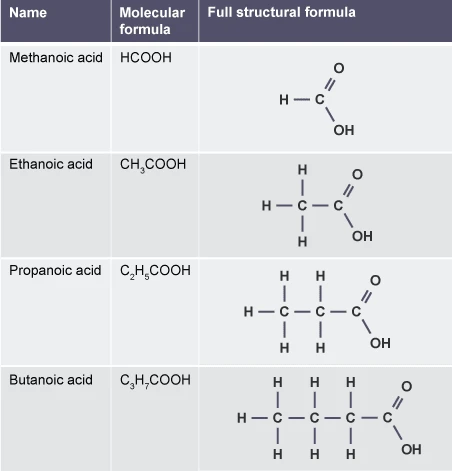
Carboxylic acids contain the functional group:
−COOH
This functional group includes:
\( \mathrm{-C(=O)OH} \)
You must be able to:
- Draw structural formulae.
- Draw displayed formulae.
- Name unbranched-chain carboxylic acids up to four carbon atoms.
Naming Rules
- Count the total number of carbon atoms (including the −COOH carbon).
- Change the suffix “-ane” to -oic acid.
- The carboxyl carbon is always carbon 1.
1. Methanoic Acid
Molecular formula:
\( \mathrm{HCOOH} \)
Structural formula:
\( \mathrm{HCOOH} \)
Displayed formula (simplified):
\( \mathrm{H – C(=O) – OH} \)
2. Ethanoic Acid
Molecular formula:
\( \mathrm{CH_3COOH} \)
Structural formula:
\( \mathrm{CH_3COOH} \)
Displayed formula (simplified):
\( \mathrm{CH_3 – C(=O) – OH} \)
3. Propanoic Acid
Molecular formula:
\( \mathrm{C_2H_5COOH} \)
Structural formula:
\( \mathrm{CH_3CH_2COOH} \)
4. Butanoic Acid
Molecular formula:
\( \mathrm{C_3H_7COOH} \)
Structural formula:
\( \mathrm{CH_3CH_2CH_2COOH} \)
| Number of Carbons | Structural Formula | Name |
|---|---|---|
| 1 | \( \mathrm{HCOOH} \) | Methanoic acid |
| 2 | \( \mathrm{CH_3COOH} \) | Ethanoic acid |
| 3 | \( \mathrm{CH_3CH_2COOH} \) | Propanoic acid |
| 4 | \( \mathrm{CH_3CH_2CH_2COOH} \) | Butanoic acid |
Key Points
- All contain the −COOH group.
- The −COOH carbon counts as part of the chain.
- Names always end in “-oic acid”.
- Only unbranched chains are required.
Example 1 (Conceptual):
Why does propanoic acid have three carbon atoms?
▶️ Answer/Explanation
The carbon in the −COOH group counts as part of the carbon chain.
So \( \mathrm{CH_3CH_2COOH} \) contains three carbon atoms in total.
Example 2 (Application):
Name the compound with structural formula \( \mathrm{CH_3CH_2CH_2COOH} \).
▶️ Answer/Explanation
There are four carbon atoms including the −COOH carbon.
The name is butanoic acid.
Example 3 (Hard):
Explain fully how you would name \( \mathrm{CH_3COOH} \).
▶️ Answer/Explanation
The molecule contains the −COOH functional group.
There are two carbon atoms including the carboxyl carbon.
The root name is ethan-.
The suffix for carboxylic acids is -oic acid.
Therefore the name is ethanoic acid.
