Edexcel iGCSE Chemistry -4.36C Reactions of Carboxylic Acids- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.36C Reactions of Carboxylic Acids- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.36C Reactions of Carboxylic Acids- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.36C describe the reactions of aqueous solutions of carboxylic acids with metals and metal carbonates
4.36C Reactions of Aqueous Carboxylic Acids with Metals and Metal Carbonates
Carboxylic acids contain the functional group:
−COOH
In aqueous solution, they behave as weak acids.
They react in a similar way to other acids such as hydrochloric acid.
1. Reaction with Metals
Carboxylic acids react with reactive metals to form:
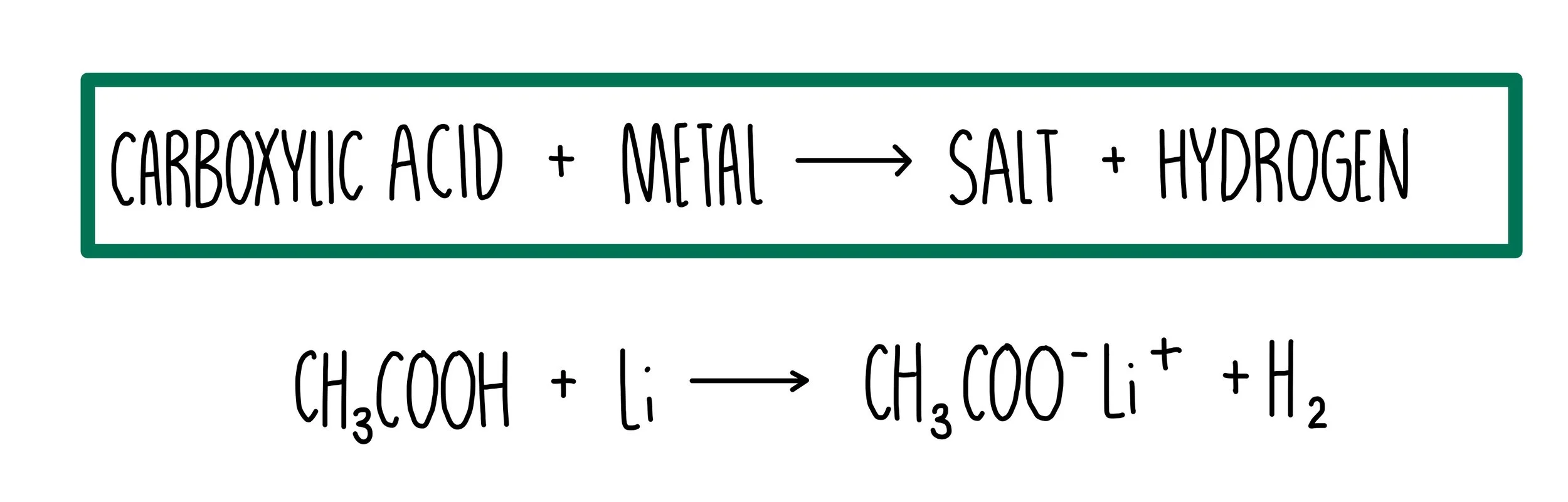
- A salt (metal carboxylate)
- Hydrogen gas
General equation:
Acid + Metal → Salt + Hydrogen
Example: Ethanoic acid and magnesium
\( \mathrm{2CH_3COOH + Mg \rightarrow (CH_3COO)_2Mg + H_2} \)
Observation:
- Effervescence (bubbles of hydrogen gas).
- Magnesium gradually disappears.
2. Reaction with Metal Carbonates
Carboxylic acids react with metal carbonates to form:
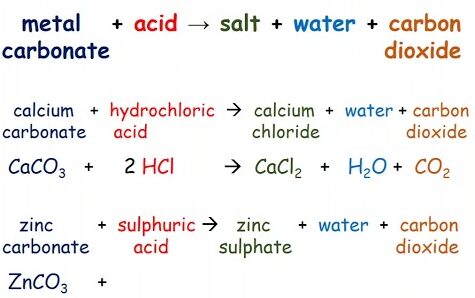
- A salt
- Carbon dioxide
- Water
General equation:
Acid + Carbonate → Salt + Carbon dioxide + Water
Example: Ethanoic acid and sodium carbonate
\( \mathrm{2CH_3COOH + Na_2CO_3 \rightarrow 2CH_3COONa + CO_2 + H_2O} \)
Observation:
- Effervescence due to carbon dioxide.
- Carbon dioxide turns limewater milky.
| Reactant | Products | Gas Produced |
|---|---|---|
| Metal | Salt + Hydrogen | \( \mathrm{H_2} \) |
| Metal carbonate | Salt + \( \mathrm{CO_2} \) + Water | \( \mathrm{CO_2} \) |
Why These Reactions Occur
- Carboxylic acids release \( \mathrm{H^+} \) ions in aqueous solution.
- Metals react with \( \mathrm{H^+} \) to produce hydrogen gas.
- Carbonates react with \( \mathrm{H^+} \) to produce carbon dioxide and water.
Key Points
- Carboxylic acids behave like other acids in aqueous solution.
- They form metal salts called carboxylates.
- Hydrogen is produced with metals.
- Carbon dioxide is produced with carbonates.
Example 1 (Conceptual):
Why is hydrogen gas produced when ethanoic acid reacts with magnesium?
▶️ Answer/Explanation
Ethanoic acid releases \( \mathrm{H^+} \) ions in solution.
Magnesium reacts with \( \mathrm{H^+} \) to form hydrogen gas.
Example 2 (Application):
Write the balanced equation for the reaction between methanoic acid and sodium carbonate.
▶️ Answer/Explanation
\( \mathrm{2HCOOH + Na_2CO_3 \rightarrow 2HCOONa + CO_2 + H_2O} \)
Example 3 (Hard):
Explain fully what would be observed when aqueous propanoic acid reacts with calcium carbonate.
▶️ Answer/Explanation
Bubbles would be seen due to carbon dioxide gas.
The calcium carbonate would gradually dissolve.
A calcium propanoate salt would form.
Water would also be produced.
The reaction occurs because the acid releases \( \mathrm{H^+} \) ions.
