Edexcel iGCSE Chemistry -4.37C Vinegar as Ethanoic Acid- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.37C Vinegar as Ethanoic Acid- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.37C Vinegar as Ethanoic Acid- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.37C know that vinegar is an aqueous solution containing ethanoic acid
4.37C Vinegar as an Aqueous Solution of Ethanoic Acid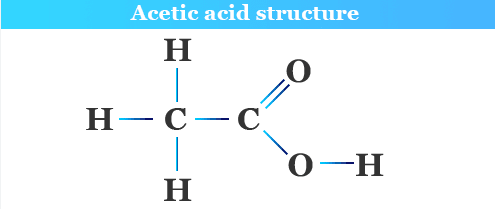
Vinegar is an aqueous solution of ethanoic acid.
“Aqueous” means dissolved in water.
Composition of Vinegar
- Contains water as the solvent.
- Contains ethanoic acid \( \mathrm{CH_3COOH} \) as the solute.
- Typically about 5–8% ethanoic acid by volume.
Ethanoic Acid in Vinegar
Ethanoic acid contains the functional group:
−COOH
In aqueous solution, it partially ionises:
\( \mathrm{CH_3COOH \rightleftharpoons H^+ + CH_3COO^-} \)
Because it only partially ionises, it is a weak acid.
How Vinegar Is Produced
Vinegar is produced by oxidation of ethanol in air:
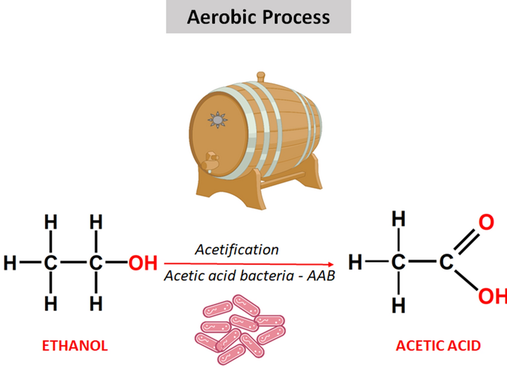
\( \mathrm{C_2H_5OH + O_2 \rightarrow CH_3COOH + H_2O} \)
This process involves bacteria and is called microbial oxidation.
| Substance | Contains Ethanoic Acid? | Acidic? |
|---|---|---|
| Water | No | Neutral |
| Vinegar | Yes | Weakly acidic |
Key Points
- Vinegar is a solution of ethanoic acid in water.
- It is an aqueous solution.
- Ethanoic acid is a weak acid.
- Ethanoic acid is produced by oxidation of ethanol.
Example 1 (Conceptual):
Why is vinegar acidic?
▶️ Answer/Explanation
It contains ethanoic acid.
Ethanoic acid releases \( \mathrm{H^+} \) ions in solution.
Example 2 (Application):
State the chemical formula of the acid present in vinegar.
▶️ Answer/Explanation
\( \mathrm{CH_3COOH} \)
Example 3 (Hard ):
Explain fully why vinegar is described as an aqueous solution containing ethanoic acid.
▶️ Answer/Explanation
A solution is formed when a solute dissolves in a solvent.
In vinegar, ethanoic acid is dissolved in water.
Water is the solvent.
Ethanoic acid is the solute.
Therefore vinegar is an aqueous solution of ethanoic acid.
