Edexcel iGCSE Chemistry -4.38C Functional Group of Esters- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.38C Functional Group of Esters- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.38C Functional Group of Esters- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.38C know that esters contain the functional group –COO–
4.38C Esters and Their Functional Group
Esters are a homologous series of organic compounds.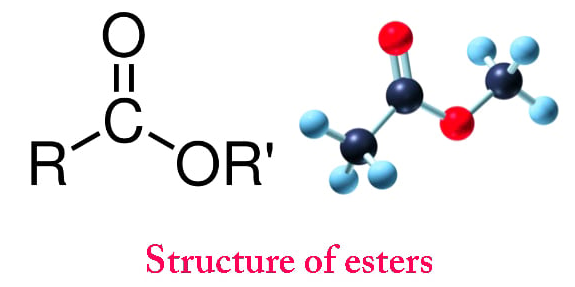
They contain the functional group:
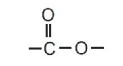
This functional group is formed from a carboxylic acid and an alcohol.
Structure of the Ester Group
The −COO− group can be represented as:
\( \mathrm{-C(=O)O-} \)
It contains:
- A carbon–oxygen double bond (C=O).
- A single C–O bond linking to another carbon chain.
General Structure of an Ester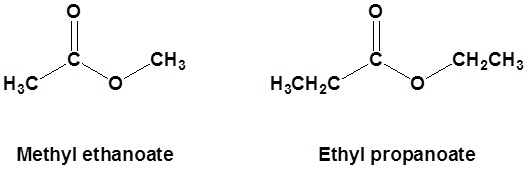
\( \mathrm{R – C(=O)O – R’} \)
Where:
- \( \mathrm{R} \) comes from the carboxylic acid.
- \( \mathrm{R’} \) comes from the alcohol.
Example
Ethyl ethanoate
\( \mathrm{CH_3COOCH_2CH_3} \)
This molecule contains the −COO− functional group.
| Compound Type | Functional Group |
|---|---|
| Alcohol | −OH |
| Carboxylic Acid | −COOH |
| Ester | −COO− |
Key Properties of Esters
- Often have pleasant fruity smells.
- Formed from alcohol + carboxylic acid.
- Contain the −COO− functional group.
- Names usually end in -oate.
Key Points
- Functional group is −COO−.
- Contains both C=O and C–O bonds.
- Formed by reaction of alcohol and carboxylic acid.
Example 1 (Conceptual):
Why is ethyl ethanoate classified as an ester?
▶️ Answer/Explanation
It contains the −COO− functional group.
This group defines esters.
Example 2 (Application):
State the functional group present in all esters.
▶️ Answer/Explanation
The −COO− functional group.
Example 3 (Hard ):
Explain fully how the functional group of an ester differs from that of a carboxylic acid.
▶️ Answer/Explanation
Carboxylic acids contain the −COOH group.
This contains a carbon–oxygen double bond and a hydroxyl group.
Esters contain the −COO− group.
In esters, the −OH of the acid is replaced by an −O− linked to another carbon chain.
Therefore esters have a different functional group from carboxylic acids.
