Edexcel iGCSE Chemistry -4.39C Formation of Ethyl Ethanoate- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.39C Formation of Ethyl Ethanoate- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.39C Formation of Ethyl Ethanoate- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.39C know that ethyl ethanoate is the ester produced when ethanol and ethanoic acid react in the presence of an acid catalyst
4.39C Formation of Ethyl Ethanoate
Ethyl ethanoate is an ester formed when:

- Ethanol \( \mathrm{C_2H_5OH} \)
- Ethanoic acid \( \mathrm{CH_3COOH} \)
react together in the presence of an acid catalyst.
The Reaction (Esterification)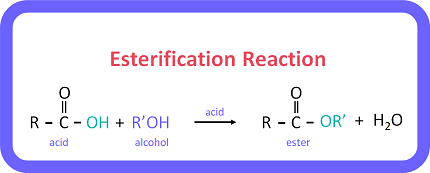
\( \mathrm{CH_3COOH + C_2H_5OH \rightleftharpoons CH_3COOC_2H_5 + H_2O} \)
This reaction is called esterification.
It is a reversible reaction.
Conditions Required
- Concentrated sulfuric acid as an acid catalyst.
- Heating under reflux.
The acid catalyst:
- Speeds up the reaction.
- Is not used up.
- Also removes water, helping the equilibrium move towards ester formation.
How the Ester Is Formed
- The −OH from the carboxylic acid combines with a hydrogen from the alcohol.
- This forms water.
- The remaining parts join together to form the ester.
| Reactants | Catalyst | Products |
|---|---|---|
| Ethanol + Ethanoic acid | Concentrated \( \mathrm{H_2SO_4} \) | Ethyl ethanoate + Water |
Properties of Ethyl Ethanoate
- Has a sweet fruity smell.
- Is a colourless liquid.
- Contains the −COO− functional group.
Key Points
- Formed from ethanol and ethanoic acid.
- Requires an acid catalyst.
- Water is produced.
- Reaction is reversible.
Example 1 (Conceptual):
Why is concentrated sulfuric acid used in the formation of ethyl ethanoate?
▶️ Answer/Explanation
It acts as an acid catalyst.
It speeds up the reaction and is not used up.
It also removes water.
Example 2 (Application):
Write the balanced equation for the formation of ethyl ethanoate.
▶️ Answer/Explanation
\( \mathrm{CH_3COOH + C_2H_5OH \rightleftharpoons CH_3COOC_2H_5 + H_2O} \)
Example 3 (Hard ):
Explain fully how ethyl ethanoate is formed from ethanol and ethanoic acid.
▶️ Answer/Explanation
Ethanol reacts with ethanoic acid in a reversible reaction.
The reaction is carried out in the presence of concentrated sulfuric acid.
The acid acts as a catalyst.
The −OH from the acid and a hydrogen from the alcohol form water.
The remaining parts join together to form the ester ethyl ethanoate.
