Edexcel iGCSE Chemistry -4.4 IUPAC Naming of Organic Compounds- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.4 IUPAC Naming of Organic Compounds- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.4 IUPAC Naming of Organic Compounds- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.4 understand how to name compounds relevant to this specification using the rules of International Union of Pure and Applied Chemistry (IUPAC) nomenclature
(students will be expected to name compounds containing up to six carbon atoms)
4.4 IUPAC Naming of Organic Compounds (Up to 6 Carbon Atoms)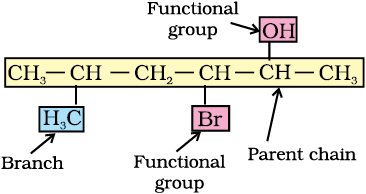
Organic compounds are named using rules set by the International Union of Pure and Applied Chemistry (IUPAC).
At GCSE level, students must name compounds containing up to six carbon atoms.
Step 1: Identify the Longest Carbon Chain
The length of the longest carbon chain determines the prefix:
| Number of Carbons | Prefix |
|---|---|
| 1 | Meth- |
| 2 | Eth- |
| 3 | Prop- |
| 4 | But- |
| 5 | Pent- |
| 6 | Hex- |
Step 2: Identify the Type of Bond
- Single bonds only → -ane (alkane)
- Double bond present → -ene (alkene)
- Contains \( \mathrm{-OH} \) group → -ol (alcohol)
Step 3: Number the Carbon Chain (If Needed)
Number the chain from the end nearest the functional group or double bond.
Example:
\( \mathrm{CH_3CH=CHCH_3} \)
The double bond starts on carbon 2 → name is but-2-ene.
Naming Examples
\( \mathrm{CH_4} \) → methane
\( \mathrm{C_2H_6} \) → ethane
\( \mathrm{C_3H_8} \) → propane
\( \mathrm{C_2H_4} \) → ethene
\( \mathrm{CH_3CH_2OH} \) → ethanol
Naming Branched Compounds (Basic Level)
Branches are named as alkyl groups (e.g. methyl \( \mathrm{CH_3} \)).
Example:
\( \mathrm{CH_3CH(CH_3)CH_3} \)
Longest chain = 3 carbons (propane) Methyl group on carbon 2 → 2-methylpropane
| Structure | Name |
|---|---|
| \( \mathrm{CH_3CH_2CH_3} \) | Propane |
| \( \mathrm{CH_2=CH_2} \) | Ethene |
| \( \mathrm{CH_3CH_2OH} \) | Ethanol |
| \( \mathrm{CH_3CH=CHCH_3} \) | But-2-ene |
Common GCSE Mistakes
- Forgetting to number the double bond.
- Choosing the wrong longest chain.
- Forgetting the “e” in alcohol names (ethanol not ethanal).
Example 1 (Conceptual):
Why is \( \mathrm{C_3H_8} \) named propane?
▶️ Answer/Explanation
It has three carbon atoms (prop-).
It contains only single bonds (-ane).
Example 2 (Application):
Name the compound \( \mathrm{CH_3CH_2CH_2CH_3} \).
▶️ Answer/Explanation
Longest chain = 4 carbons → but-
All single bonds → -ane
Name = butane
Example 3 (Hard):
Explain fully why \( \mathrm{CH_3CH(OH)CH_3} \) is named propan-2-ol.
▶️ Answer/Explanation
The longest carbon chain contains three carbons → prop-.
The molecule contains an alcohol functional group \( \mathrm{-OH} \) → -ol.
The \( \mathrm{-OH} \) group is attached to carbon 2.
Therefore the correct name is propan-2-ol.
