Edexcel iGCSE Chemistry -4.40C Structure of Ethyl Ethanoate- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.40C Structure of Ethyl Ethanoate- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.40C Structure of Ethyl Ethanoate- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.40C understand how to write the structural and displayed formulae of ethyl ethanoate
4.40C Structural and Displayed Formulae of Ethyl Ethanoate
Ethyl ethanoate is an ester formed from: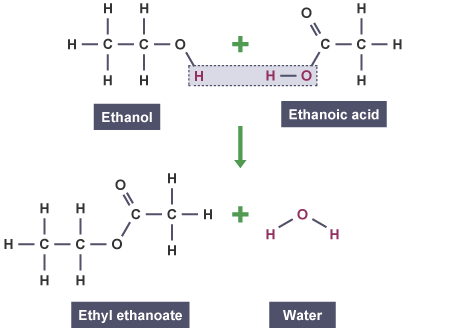
- Ethanoic acid \( \mathrm{CH_3COOH} \)
- Ethanol \( \mathrm{C_2H_5OH} \)
It contains the functional group:
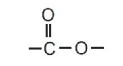
Molecular Formula
\( \mathrm{C_4H_8O_2} \)
Structural Formula
The condensed (structural) formula is:
\( \mathrm{CH_3COOCH_2CH_3} \)
This shows:
- The \( \mathrm{CH_3CO-} \) part from ethanoic acid.
- The \( \mathrm{-OCH_2CH_3} \) part from ethanol.
Displayed Formula
The displayed formula must show:
- The carbon–oxygen double bond.
- The C–O single bond linking to the ethyl group.
- All atoms and covalent bonds.
\( \mathrm{CH_3 – C(=O) – O – CH_2 – CH_3} \)
This clearly shows the ester functional group \( \mathrm{-C(=O)O-} \).
| Type of Formula | Representation |
|---|---|
| Structural formula | \( \mathrm{CH_3COOCH_2CH_3} \) |
| Displayed formula | \( \mathrm{CH_3 – C(=O) – O – CH_2 – CH_3} \) |
How to Construct the Formula
- Take the carboxylic acid structure \( \mathrm{CH_3COOH} \).
- Remove the −OH from the acid.
- Take ethanol \( \mathrm{C_2H_5OH} \).
- Remove the hydrogen from its −OH group.
- Join the remaining parts with an −O− linkage.
Exam Key Points
- Must include C=O double bond.
- Must show the −COO− linkage.
- Ethyl part contains two carbon atoms.
- Ethanoate part contains the C=O group.
Example 1 (Conceptual):
Why must the displayed formula of ethyl ethanoate show a C=O double bond?
▶️ Answer/Explanation
The ester functional group contains a carbon–oxygen double bond.
Without it, the molecule would not be an ester.
Example 2 (Application):
Write the structural formula of ethyl ethanoate.
▶️ Answer/Explanation
\( \mathrm{CH_3COOCH_2CH_3} \)
Example 3 (Hard):
Explain fully how you would construct the displayed formula of ethyl ethanoate from ethanol and ethanoic acid.
▶️ Answer/Explanation
Start with ethanoic acid \( \mathrm{CH_3COOH} \).
Remove the −OH from the acid.
Take ethanol \( \mathrm{C_2H_5OH} \).
Remove the hydrogen from its −OH group.
Join the remaining parts with an oxygen atom.
Include the carbon–oxygen double bond in the acid part.
The final structure is \( \mathrm{CH_3 – C(=O) – O – CH_2 – CH_3} \).
