Edexcel iGCSE Chemistry -4.41–4.42C Structures of Esters , Properties and Uses of Esters- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.41–4.42C Structures of Esters , Properties and Uses of Esters- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.41–4.42C Structures of Esters , Properties and Uses of Esters- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.41C understand how to write the structural and displayed formulae of an ester, given the name or formula of the alcohol and carboxylic acid from which it is formed and vice versa
4.42C know that esters are volatile compounds with distinctive smells and are used as food flavourings and in perfumes
4.41C Writing Structural and Displayed Formulae of Esters from Alcohols and Carboxylic Acids (and Vice Versa)
Esters are formed by the reaction between: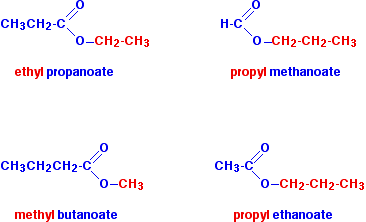
- An alcohol (contains −OH)
- A carboxylic acid (contains −COOH)
This reaction is called esterification.
\( \mathrm{Carboxylic\ acid + Alcohol \rightleftharpoons Ester + Water} \)
The Ester Functional Group
\( \mathrm{-C(=O)O-} \)
When forming an ester:
- The −OH from the acid is removed.
- The H from the alcohol’s −OH is removed.
- These combine to form water.
- The remaining parts join through an −O− link.
Step-by-Step Method (From Acid + Alcohol to Ester)
Example: Ethanoic acid + Propanol
Ethanoic acid:
\( \mathrm{CH_3COOH} \)
Propan-1-ol:
\( \mathrm{CH_3CH_2CH_2OH} \)
Remove:
- −OH from the acid
- H from the alcohol
Join remaining parts:
\( \mathrm{CH_3COOCH_2CH_2CH_3} \)
Name: propyl ethanoate
Naming Rule for Esters
- The alcohol gives the first part of the name (alkyl group).
- The acid gives the second part (ending changes to -oate).
Example:
- Ethanol → Ethyl
- Ethanoic acid → Ethanoate
- Product → Ethyl ethanoate
Working Backwards (From Ester to Acid + Alcohol)
Example: Ethyl propanoate
- “Ethyl” comes from ethanol
- “Propanoate” comes from propanoic acid
Alcohol:
\( \mathrm{C_2H_5OH} \)
Acid:
\( \mathrm{CH_3CH_2COOH} \)
| Ester Name | Alcohol | Carboxylic Acid |
|---|---|---|
| Ethyl ethanoate | Ethanol | Ethanoic acid |
| Propyl ethanoate | Propanol | Ethanoic acid |
| Ethyl propanoate | Ethanol | Propanoic acid |
Displayed Formula Example
Propyl ethanoate:
\( \mathrm{CH_3 – C(=O) – O – CH_2 – CH_2 – CH_3} \)
Must clearly show:
- The C=O double bond.
- The −O− linkage.
- All atoms.
Key Points
- Alcohol part becomes the first name.
- Acid part changes to -oate.
- Remove −OH from acid and H from alcohol.
- Join through −O−.
Example 1 (Conceptual):
From which alcohol and acid is methyl ethanoate formed?
▶️ Answer/Explanation
Methyl comes from methanol.
Ethanoate comes from ethanoic acid.
Example 2 (Application):
Write the structural formula of ethyl propanoate.
▶️ Answer/Explanation
Propanoate part: \( \mathrm{CH_3CH_2COO-} \)
Ethyl part: \( \mathrm{-CH_2CH_3} \)
\( \mathrm{CH_3CH_2COOCH_2CH_3} \)
Example 3 (Hard):
Explain fully how you would determine the alcohol and carboxylic acid used to form butyl ethanoate.
▶️ Answer/Explanation
The first part “butyl” comes from butanol.
The second part “ethanoate” comes from ethanoic acid.
So the alcohol is butanol.
The acid is ethanoic acid.
The ester contains the −COO− functional group linking the two parts.
4.42C Properties and Uses of Esters
Esters are organic compounds that contain the functional group:
−COO−
Physical Properties of Esters
- Esters are volatile compounds.
- Volatile means they evaporate easily at room temperature.
- They have low boiling points compared to carboxylic acids.
- They have distinctive fruity smells.
Because they evaporate easily, their smell can be detected quickly.
Why Esters Are Volatile
- They have simple molecular structures.
- They have relatively weak intermolecular forces.
- Less energy is needed to separate the molecules.
Uses of Esters
- Food flavourings
- Perfumes
- Fragrances
Different esters have different characteristic smells.
Examples:
- Ethyl ethanoate – sweet smell
- Pentyl ethanoate – banana smell
- Methyl butanoate – apple smell
| Property | Reason | Use |
|---|---|---|
| Volatile | Weak intermolecular forces | Smell spreads easily |
| Fruity smell | Specific molecular structure | Flavourings & perfumes |
Key Points
- Esters are volatile.
- They have distinctive fruity smells.
- Used in flavourings and perfumes.
- Contain the −COO− functional group.
Example 1 (Conceptual):
Why can the smell of an ester be detected quickly?
▶️ Answer/Explanation
Esters are volatile.
They evaporate easily and spread through the air.
Example 2 (Application):
State two uses of esters.
▶️ Answer/Explanation
Food flavourings.
Perfumes.
Example 3 (Hard ):
Explain fully why esters are suitable for use in perfumes.
▶️ Answer/Explanation
Esters are volatile so they evaporate easily.
This allows their smell to spread into the air.
They have distinctive pleasant fruity smells.
Therefore they are suitable for perfumes.
