Edexcel iGCSE Chemistry -4.43C Practical: Preparation of an Ester- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.43C Practical: Preparation of an Ester- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.43C Practical: Preparation of an Ester- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.43C practical: prepare a sample of an ester such as ethyl ethanoate
4.43C Practical: Preparation of an Ester (e.g. Ethyl Ethanoate)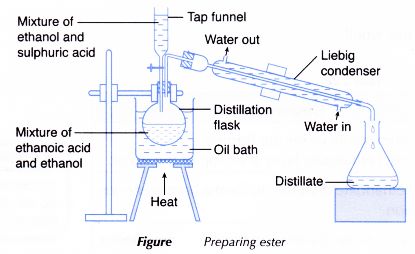
In this practical, an ester such as ethyl ethanoate is prepared by reacting:
- Ethanoic acid \( \mathrm{CH_3COOH} \)
- Ethanol \( \mathrm{C_2H_5OH} \)
in the presence of an acid catalyst.
The Reaction (Esterification)
\( \mathrm{CH_3COOH + C_2H_5OH \rightleftharpoons CH_3COOC_2H_5 + H_2O} \)
The reaction is reversible and requires an acid catalyst.
Apparatus Required
- Test tube or round-bottom flask

- Measuring cylinder
- Water bath
- Beaker
- Separating funnel (optional)
Chemicals Required
- Ethanol
- Ethanoic acid
- Concentrated sulfuric acid \( \mathrm{H_2SO_4} \) (catalyst)
- Sodium carbonate solution (to neutralise excess acid)
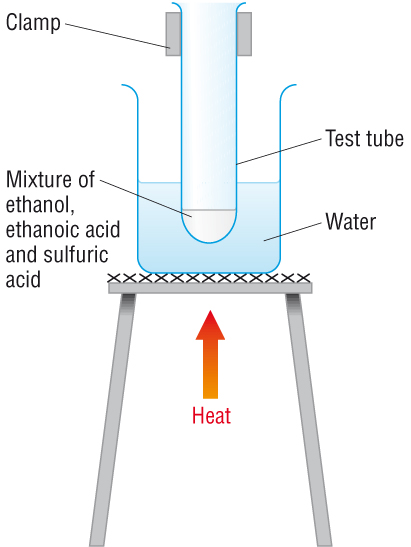
Method
- Measure equal volumes of ethanol and ethanoic acid into a test tube.
- Add a few drops of concentrated sulfuric acid carefully.
- Place the test tube in a hot water bath for several minutes.
- Pour the mixture into a beaker containing sodium carbonate solution.
- A sweet fruity smell indicates the ester has formed.
Observations
- Fruity smell of ethyl ethanoate.
- Effervescence when poured into sodium carbonate (due to neutralisation).
| Step | Purpose |
|---|---|
| Add sulfuric acid | Acts as catalyst and removes water |
| Heat in water bath | Increases reaction rate safely |
| Add sodium carbonate | Neutralises excess acid |
Safety Precautions
- Wear eye protection.
- Handle concentrated sulfuric acid carefully.
- Heat using a water bath, not a direct flame (ethanol is flammable).
Why a Water Bath Is Used
- Ethanol is flammable.
- A direct flame could cause ignition.
- A water bath provides gentle and safe heating.
Example 1 (Conceptual):
Why must the reaction mixture be heated in a water bath rather than over a flame?
▶️ Answer/Explanation
Ethanol is flammable.
A direct flame could cause it to catch fire.
Example 2 (Application):
State the role of concentrated sulfuric acid in this practical.
▶️ Answer/Explanation
It acts as an acid catalyst.
It also removes water and speeds up the reaction.
Example 3 (Hard):
Explain fully why sodium carbonate solution is added after heating the reaction mixture.
▶️ Answer/Explanation
Sodium carbonate neutralises any excess ethanoic acid and sulfuric acid.
This stops further reaction.
It also reduces the acidic smell so the fruity smell of the ester can be detected.