Edexcel iGCSE Chemistry -4.44–4.45 Addition Polymerisation and its Repeated Units- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.44–4.45 Addition Polymerisation and its Repeated Units- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.44–4.45 Addition Polymerisation and its Repeated Units- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.44 know that an addition polymer is formed by joining up many small molecules called monomers
4.45 understand how to draw the repeat unit of an addition polymer, including poly(ethene), poly(propene), poly(chloroethene) and (poly)tetrafluoroethene
4.44 Addition Polymers and Monomers
An addition polymer is formed when many small molecules called monomers join together to make one very large molecule.
This process is called addition polymerisation.
Monomer
A monomer is a small molecule that can join to other similar molecules.
In addition polymerisation, the monomer usually contains a carbon–carbon double bond.
Example monomer: ethene \( \mathrm{CH_2=CH_2} \)
Polymer
A polymer is a very large molecule made from repeating units of monomers.
When ethene polymerises:
\( \mathrm{nCH_2=CH_2 \rightarrow [-CH_2-CH_2-]_n} \)
The double bond opens and forms long chains.
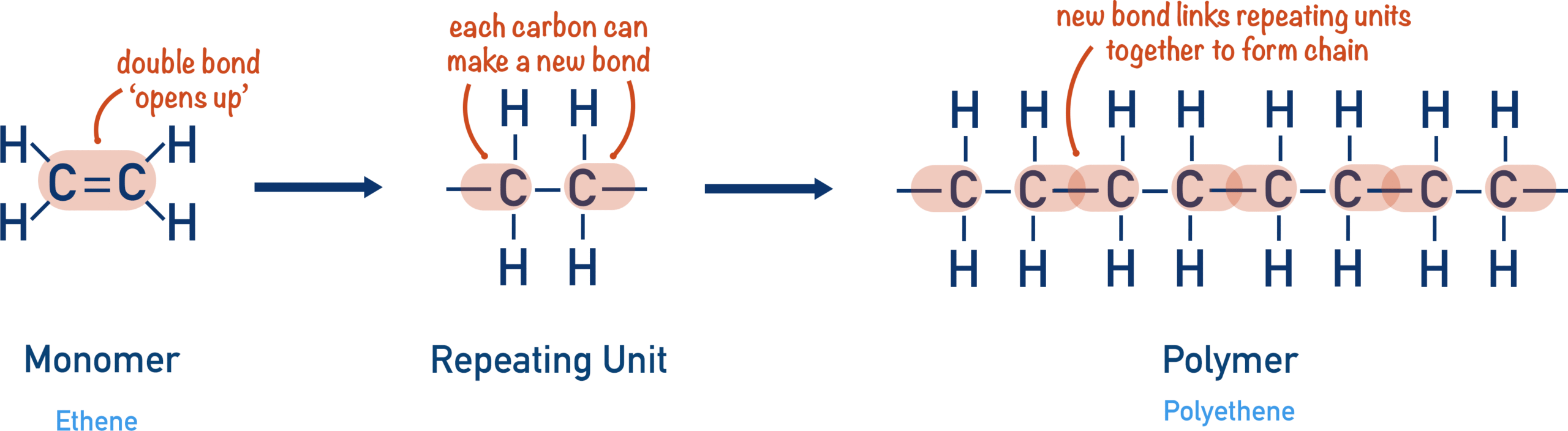
Why It Is Called “Addition” Polymerisation
- The monomers join together.
- No other products are formed.
- All atoms from the monomers are present in the polymer.
| Term | Definition |
|---|---|
| Monomer | Small molecule that joins to others |
| Polymer | Large molecule made of repeating units |
| Addition polymer | Formed by addition polymerisation |
Key GCSE Points
- Addition polymers form from alkenes.
- The C=C double bond opens.
- No small molecules are eliminated.
- The polymer is made from repeating units.
Example 1 (Conceptual):
Why must the monomer contain a carbon–carbon double bond?
▶️ Answer/Explanation
The double bond can open.
This allows the monomers to join into long chains.
Example 2 (Application):
Write the repeating unit for the polymer formed from ethene.
▶️ Answer/Explanation
\( \mathrm{[-CH_2-CH_2-]_n} \)
Example 3 (Hard):
Explain fully why addition polymerisation does not produce any small molecules as by-products.
▶️ Answer/Explanation
In addition polymerisation, the double bond opens.
The monomers join directly to each other.
No atoms are removed from the monomer.
Therefore no small molecules are formed as by-products.
4.45 Drawing the Repeat Unit of Addition Polymers
An addition polymer is formed when many alkene monomers join together.
To draw the repeat unit:
- Start with the alkene monomer.
- Open the C=C double bond.
- Convert it into a single bond.
- Place brackets around the repeating section.
- Add a small \( \mathrm{n} \) outside the brackets.
1. Poly(ethene)
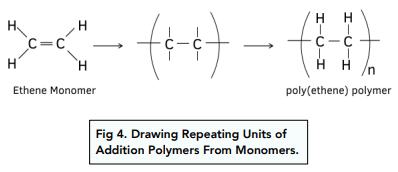
Monomer: Ethene
\( \mathrm{CH_2=CH_2} \)
Repeat unit:
\( \mathrm{[-CH_2-CH_2-]_n} \)
2. Poly(propene)
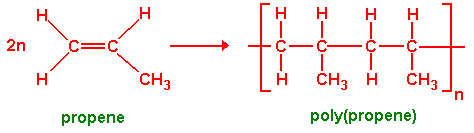
Monomer: Propene
\( \mathrm{CH_2=CHCH_3} \)
Repeat unit:
\( \mathrm{[-CH_2-CH(CH_3)-]_n} \)
The \( \mathrm{CH_3} \) group stays attached to the second carbon.
3. Poly(chloroethene) (PVC)
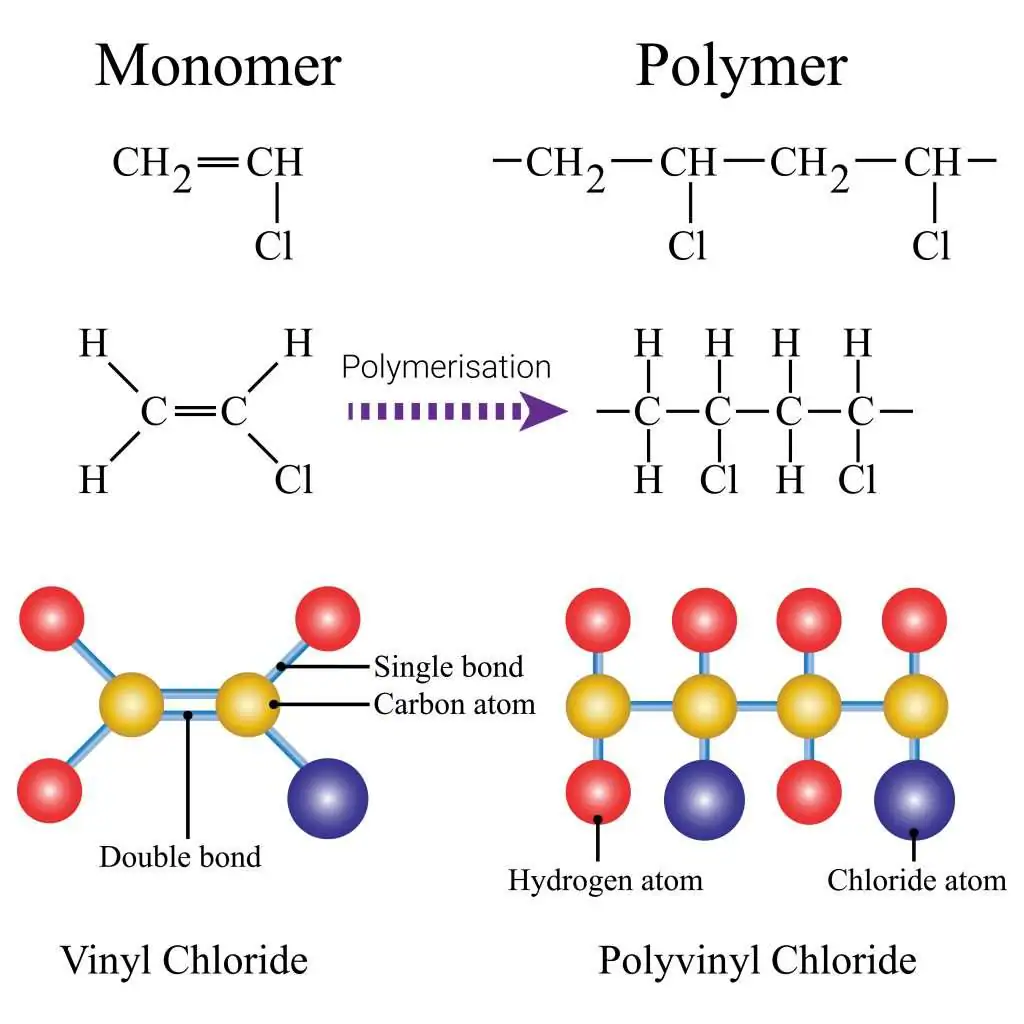
Monomer: Chloroethene
\( \mathrm{CH_2=CHCl} \)
Repeat unit:
\( \mathrm{[-CH_2-CH(Cl)-]_n} \)
The chlorine atom remains bonded to the second carbon.
4. Poly(tetrafluoroethene) (PTFE)
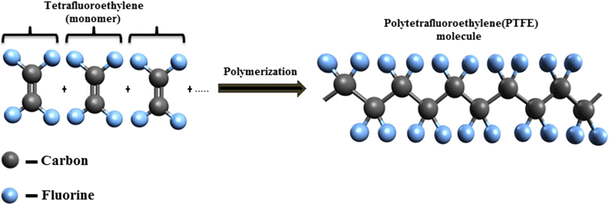
Monomer: Tetrafluoroethene
\( \mathrm{CF_2=CF_2} \)
Repeat unit:
\( \mathrm{[-CF_2-CF_2-]_n} \)
| Polymer | Monomer | Repeat Unit |
|---|---|---|
| Poly(ethene) | \( \mathrm{CH_2=CH_2} \) | \( \mathrm{[-CH_2-CH_2-]_n} \) |
| Poly(propene) | \( \mathrm{CH_2=CHCH_3} \) | \( \mathrm{[-CH_2-CH(CH_3)-]_n} \) |
| Poly(chloroethene) | \( \mathrm{CH_2=CHCl} \) | \( \mathrm{[-CH_2-CH(Cl)-]_n} \) |
| Poly(tetrafluoroethene) | \( \mathrm{CF_2=CF_2} \) | \( \mathrm{[-CF_2-CF_2-]_n} \) |
Common Exam Mistakes
- Forgetting to remove the double bond.
- Forgetting brackets around the repeat unit.
- Placing \( \mathrm{n} \) inside the brackets instead of outside.
Key Points
- Open the C=C bond.
- Keep side groups attached to the same carbon.
- Use brackets and write \( \mathrm{n} \) outside.
Example 1 (Conceptual):
Why does the double bond disappear in the repeat unit?
▶️ Answer/Explanation
The double bond opens during polymerisation.
It becomes a single bond linking monomers together.
Example 2 (Application):
Draw the repeat unit of poly(chloroethene).
▶️ Answer/Explanation
\( \mathrm{[-CH_2-CH(Cl)-]_n} \)
Example 3 (Hard):
Explain fully how you would draw the repeat unit of poly(propene) from its monomer.
▶️ Answer/Explanation
Start with the monomer \( \mathrm{CH_2=CHCH_3} \).
Open the carbon–carbon double bond.
Replace it with a single bond.
Keep the \( \mathrm{CH_3} \) group attached to the second carbon.
Place brackets around the repeating section.
Write \( \mathrm{n} \) outside the brackets.
