Edexcel iGCSE Chemistry -4.46 Deducing Monomers from Polymers- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.46 Deducing Monomers from Polymers- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.46 Deducing Monomers from Polymers- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.46 understand how to deduce the structure of a monomer from the repeat unit of an addition polymer and vice versa
4.46 Deducing the Structure of a Monomer from the Repeat Unit (and Vice Versa)
Addition polymers are formed from alkene monomers.
You must be able to:
- Deduced the repeat unit from a given monomer.
- Deduced the monomer from a given repeat unit.
A. From Monomer → Repeat Unit
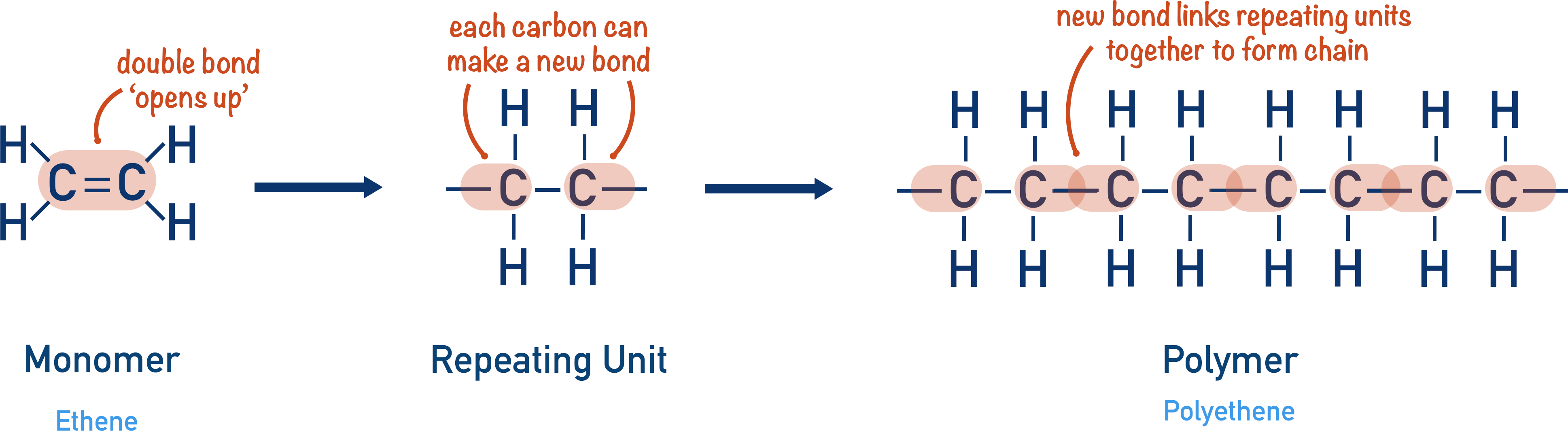
Steps:
- Identify the C=C double bond in the monomer.
- Open the double bond.
- Replace it with a single bond.
- Place brackets around the repeating section.
- Write \( \mathrm{n} \) outside the brackets.
Example: Propene
Monomer: \( \mathrm{CH_2=CHCH_3} \)
Repeat unit: \( \mathrm{[-CH_2-CH(CH_3)-]_n} \)
B. From Repeat Unit → Monomer
Steps:
- Remove the brackets and the \( \mathrm{n} \).
- Insert a double bond between the two carbon atoms in the backbone.
- Keep all side groups on the same carbon.
Example:
Repeat unit: \( \mathrm{[-CH_2-CH(Cl)-]_n} \)
Monomer: \( \mathrm{CH_2=CHCl} \)
| Given | What to Do | Result |
|---|---|---|
| Monomer | Open double bond | Repeat unit |
| Repeat unit | Insert double bond | Monomer |
Key Rule
Only addition polymers come from monomers with a C=C double bond.
Common Exam Mistakes
- Putting the double bond in the wrong place.
- Changing the position of side groups.
- Forgetting that the backbone contains two carbons.
Example 1 (Conceptual):
Why must the monomer of an addition polymer contain a C=C bond?
▶️ Answer/Explanation
The C=C bond can open.
This allows monomers to link into long chains.
Example 2 (Application):
Deduce the monomer from the repeat unit \( \mathrm{[-CF_2-CF_2-]_n} \).
▶️ Answer/Explanation
Insert a double bond between the carbons:
\( \mathrm{CF_2=CF_2} \)
The monomer is tetrafluoroethene.
Example 3 (Hard):
Explain fully how you would deduce the monomer from the repeat unit \( \mathrm{[-CH_2-CH(CH_3)-]_n} \).
▶️ Answer/Explanation
Remove the brackets and the \( \mathrm{n} \).
Identify the two-carbon backbone.
Insert a double bond between these two carbons.
Keep the \( \mathrm{CH_3} \) group attached to the second carbon.
The monomer is \( \mathrm{CH_2=CHCH_3} \), which is propene.
