Edexcel iGCSE Chemistry -4.48C Condensation Polymerisation- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.48C Condensation Polymerisation- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.48C Condensation Polymerisation- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.48C know that condensation polymerisation, in which a dicarboxylic acid reacts with a diol, produces a polyester and water
4.48C Condensation Polymerisation – Formation of Polyesters
Condensation polymerisation is a reaction in which:
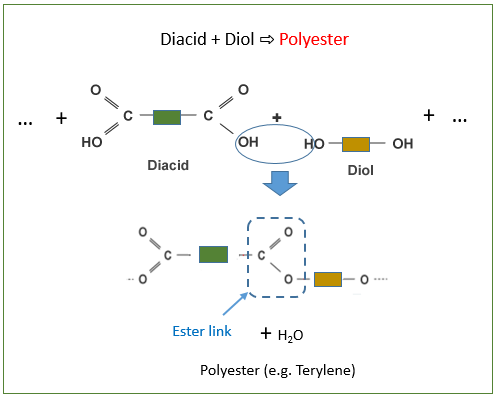
- A dicarboxylic acid
- Reacts with a diol
to form a polyester and water.
Definitions
- Dicarboxylic acid – a molecule with two \( \mathrm{-COOH} \) groups.
- Diol – a molecule with two \( \mathrm{-OH} \) groups.
- Polyester – a polymer containing many ester linkages \( \mathrm{-COO-} \).
General Reaction
Each time a link forms:
- An ester bond \( \mathrm{-COO-} \) is produced.
- One molecule of water is eliminated.
\( \mathrm{-COOH + HO- \rightarrow -COO- + H_2O} \)
Why It Is Called “Condensation”
- A small molecule is removed.
- In this case, water \( \mathrm{H_2O} \).
This is different from addition polymerisation, where no small molecules are formed.
Example Structure (Simplified)
Ethane-1,2-diol:
\( \mathrm{HO-CH_2-CH_2-OH} \)
Hexanedioic acid:
\( \mathrm{HOOC-(CH_2)_4-COOH} \)
Polymer repeat unit (simplified):
\( \mathrm{[-O-CH_2-CH_2-O-CO-(CH_2)_4-CO-]_n} \)
| Polymerisation Type | Monomers | By-product |
|---|---|---|
| Addition | Alkene | None |
| Condensation | Dicarboxylic acid + Diol | \( \mathrm{H_2O} \) |
GCSE Points
- Two different monomers are needed.
- Water is produced.
- The polymer contains ester linkages.
- The polymer formed is called a polyester.
Example 1 (Conceptual):
Why is water produced during condensation polymerisation?
▶️ Answer/Explanation
The −OH from the acid and a hydrogen from the diol combine.
This forms water.
Example 2 (Application):
State the two types of monomers needed to form a polyester.
▶️ Answer/Explanation
A dicarboxylic acid and a diol.
Example 3 (Hard ):
Explain fully how a polyester is formed from a dicarboxylic acid and a diol.
▶️ Answer/Explanation
The dicarboxylic acid contains two −COOH groups.
The diol contains two −OH groups.
An ester bond forms between −COOH and −OH groups.
Water is eliminated each time a bond forms.
This process repeats many times to produce a long polymer chain called a polyester.
