Edexcel iGCSE Chemistry -4.49C Polyesters and Their Formation- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.49C Polyesters and Their Formation- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.49C Polyesters and Their Formation- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.49C understand how to write the structural and displayed formula of a polyester, showing the repeat unit, given the formulae of the monomers from which it is formed (including ethanedioic acid and ethanediol)
4.49C Writing the Structural and Displayed Formula of a Polyester (Repeat Unit)
In condensation polymerisation, a dicarboxylic acid reacts with a diol to form a polyester and water.
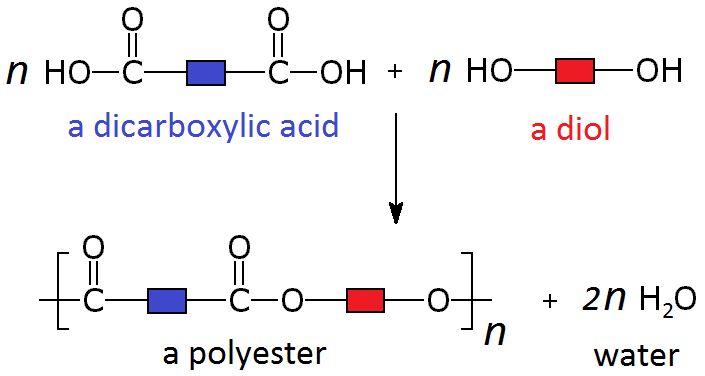
Step 1: Identify the Functional Groups
- Dicarboxylic acid → contains two \( \mathrm{-COOH} \) groups.
- Diol → contains two \( \mathrm{-OH} \) groups.
General condensation step:
\( \mathrm{-COOH + HO- \rightarrow -COO- + H_2O} \)
Each ester linkage formed produces one molecule of water.
Example Required by Specification
Ethanedioic acid:
\( \mathrm{HOOC-COOH} \)
Ethanediol:
\( \mathrm{HO-CH_2-CH_2-OH} \)
Step 2: Form the Ester Linkages
- Remove −OH from the acid.
- Remove H from the alcohol.
- Join through an −O− bond.
- Repeat at both ends.
Structural Formula of the Repeat Unit
\( \mathrm{[-O-CH_2-CH_2-O-CO-CO-]_n} \)
This shows:
- The diol section \( \mathrm{-O-CH_2-CH_2-O-} \)
- The dicarboxylic acid section \( \mathrm{-CO-CO-} \)
- The repeating ester linkage \( \mathrm{-COO-} \)
Displayed Formula of the Repeat Unit
\( \mathrm{[-O-CH_2-CH_2-O-C(=O)-C(=O)-]_n} \)
The displayed formula must clearly show:
- The C=O double bonds.
- The ester link \( \mathrm{-C(=O)O-} \).
- Brackets with \( \mathrm{n} \) outside.
| Monomer | Functional Groups | Contribution to Polymer |
|---|---|---|
| Ethanedioic acid | Two \( \mathrm{-COOH} \) | \( \mathrm{-CO-CO-} \) |
| Ethanediol | Two \( \mathrm{-OH} \) | \( \mathrm{-O-CH_2-CH_2-O-} \) |
Key Rules for Exam Questions
- Always include brackets.
- Put \( \mathrm{n} \) outside the brackets.
- Show the C=O double bonds.
- Ensure ester linkage is correct.
Example 1 (Conceptual):
Why is a polyester formed when ethanedioic acid reacts with ethanediol?
▶️ Answer/Explanation
Ethanedioic acid has two −COOH groups.
Ethanediol has two −OH groups.
Each pair reacts to form ester links and water.
This repeats to form a long chain polymer.
Example 2 (Application):
Write the structural formula of the repeat unit formed from ethanedioic acid and ethanediol.
▶️ Answer/Explanation
\( \mathrm{[-O-CH_2-CH_2-O-CO-CO-]_n} \)
Example 3 (Hard):
Explain fully how to construct the displayed formula of a polyester formed from a dicarboxylic acid and a diol.
▶️ Answer/Explanation
Identify the −COOH groups on the acid and the −OH groups on the diol.
Remove −OH from the acid and H from the alcohol.
Join them with an −O− linkage.
Include the C=O double bonds in the structure.
Repeat this linking pattern to form the repeat unit.
Place brackets around the repeating section and write \( \mathrm{n} \) outside.
