Edexcel iGCSE Chemistry -4.5 Structural and Displayed Formulae- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.5 Structural and Displayed Formulae- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.5 Structural and Displayed Formulae- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.5 understand how to write the possible structural and displayed formulae of an organic molecule given its molecular formula
4.5 Writing Possible Structural and Displayed Formulae from a Molecular Formula
The molecular formula shows the number of each type of atom in a molecule.
However, different structural arrangements may be possible. These different arrangements are called structural isomers.
Step 1: Count Carbon and Hydrogen Atoms
Use the molecular formula to determine:
- The number of carbon atoms.
- The number of hydrogen atoms.
- Whether the formula fits an alkane, alkene or alcohol pattern.
Example:
\( \mathrm{C_4H_{10}} \)
Matches the alkane general formula \( \mathrm{C_{n}H_{2n+2}} \).
Step 2: Draw All Possible Carbon Arrangements
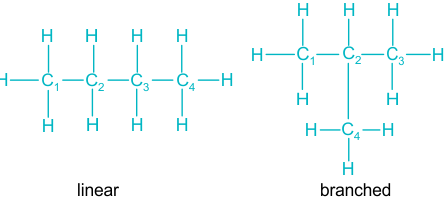
For \( \mathrm{C_4H_{10}} \), two structural possibilities exist.
Structural Formula 1:
\( \mathrm{CH_3CH_2CH_2CH_3} \) (Butane)
Structural Formula 2:
\( \mathrm{CH_3CH(CH_3)CH_3} \) (2-methylpropane)
These are structural isomers.
Displayed Formula
A displayed formula shows all atoms and all bonds.
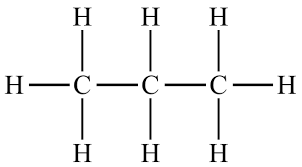
Example: \( \mathrm{C_3H_8} \)
Example: \( \mathrm{C_3H_6} \)
Matches alkene general formula \( \mathrm{C_{n}H_{2n}} \).
Structural formula:
\( \mathrm{CH_2=CHCH_3} \) (Propene)
Double bond must be shown in displayed formula.
| Molecular Formula | Possible Structures | Number of Isomers |
|---|---|---|
| \( \mathrm{C_2H_6} \) | Ethane only | 1 |
| \( \mathrm{C_3H_8} \) | Propane only | 1 |
| \( \mathrm{C_4H_{10}} \) | Butane and 2-methylpropane | 2 |
Exam Points
- Carbon forms 4 covalent bonds.
- Hydrogen forms 1 covalent bond.
- All bonds must be satisfied in displayed formula.
- Check hydrogen count carefully.
Example 1 (Conceptual):
Why can \( \mathrm{C_4H_{10}} \) form more than one structure?
▶️ Answer/Explanation
The carbon atoms can be arranged in different ways.
Both arrangements satisfy carbon’s four bonds.
Therefore structural isomers exist.
Example 2 (Application):
Draw the structural formula of one isomer of \( \mathrm{C_4H_{10}} \).
▶️ Answer/Explanation
One possible answer:
\( \mathrm{CH_3CH_2CH_2CH_3} \)
Example 3 (Hard):
A compound has the molecular formula \( \mathrm{C_4H_8} \). Explain fully how you would determine possible structures.
▶️ Answer/Explanation
The formula \( \mathrm{C_4H_8} \) matches the general formula \( \mathrm{C_{n}H_{2n}} \).
This suggests an alkene.
Draw the longest chain of four carbon atoms.
The double bond can be placed in different positions.
Possible structures include but-1-ene and but-2-ene.
Branched forms may also exist.
Each structure must satisfy carbon’s four bonds.
