Edexcel iGCSE Chemistry -4.6 Organic Reaction Types- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.6 Organic Reaction Types- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.6 Organic Reaction Types- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.6 understand how to classify reactions of organic compounds as substitution, addition and combustion
(knowledge of reaction mechanisms is not required)
4.6 Classification of Organic Reactions: Substitution, Addition and Combustion
Organic reactions can be classified into three main types:
- Substitution
- Addition
- Combustion
At GCSE level, knowledge of reaction mechanisms is not required.
1. Substitution Reactions
A substitution reaction is when one atom or group of atoms in a molecule is replaced by another atom or group.
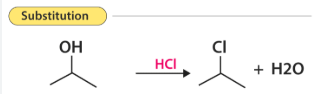
Common in alkanes.
Example: Reaction of methane with chlorine in UV light:
\( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
One hydrogen atom is replaced by a chlorine atom.
2. Addition Reactions
An addition reaction occurs when atoms add across a double bond.
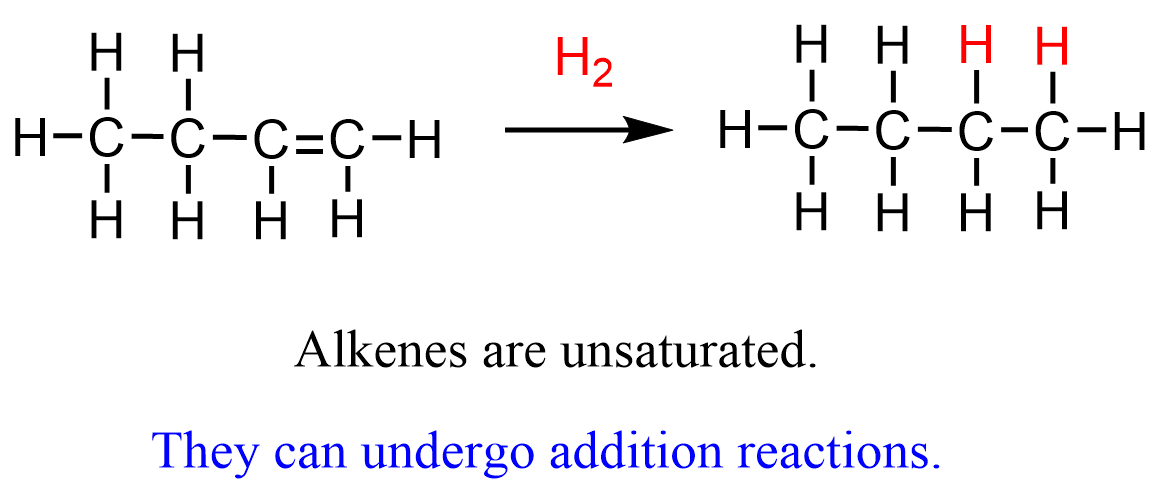
Common in alkenes.
Example: Reaction of ethene with hydrogen:
\( \mathrm{C_2H_4 + H_2 \rightarrow C_2H_6} \)
The double bond breaks and hydrogen atoms add to the carbons.
Example with bromine:
\( \mathrm{C_2H_4 + Br_2 \rightarrow C_2H_4Br_2} \)
Bromine water is decolourised (orange → colourless).
3. Combustion Reactions
A combustion reaction is when a substance reacts with oxygen to release heat energy.

Complete combustion of hydrocarbons produces carbon dioxide and water.
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Incomplete combustion can produce carbon monoxide:
\( \mathrm{2CH_4 + 3O_2 \rightarrow 2CO + 4H_2O} \)
| Reaction Type | What Happens | Example |
|---|---|---|
| Substitution | Atom replaced | \( \mathrm{CH_4 + Cl_2} \) |
| Addition | Atoms add across C=C | \( \mathrm{C_2H_4 + H_2} \) |
| Combustion | Reaction with oxygen | \( \mathrm{CH_4 + O_2} \) |
How to Identify the Reaction Type in Exams
- If a double bond disappears → addition.
- If a hydrogen is replaced by another atom → substitution.
- If oxygen is a reactant and heat released → combustion.
Example 1 (Conceptual):
Why is the reaction between ethene and bromine an addition reaction?
▶️ Answer/Explanation
The C=C double bond breaks.
Two bromine atoms add across the double bond.
No atoms are removed.
Example 2 (Application):
Classify the reaction:
\( \mathrm{C_3H_8 + Cl_2 \rightarrow C_3H_7Cl + HCl} \)
▶️ Answer/Explanation
This is a substitution reaction.
A hydrogen atom has been replaced by chlorine.
Example 3 (Hard ):
Explain fully why the reaction between propene and hydrogen is classified as an addition reaction rather than substitution.
▶️ Answer/Explanation
Propene contains a C=C double bond.
During the reaction, the double bond breaks.
Hydrogen atoms add to both carbon atoms.
No atom is replaced or removed.
The product is a saturated alkane.
Therefore it is an addition reaction.
