Edexcel iGCSE Chemistry -4.7 Crude Oil as a Mixture of Hydrocarbons- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.7 Crude Oil as a Mixture of Hydrocarbons- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.7 Crude Oil as a Mixture of Hydrocarbons- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.7 know that crude oil is a mixture of hydrocarbons
4.7 Crude Oil
Crude oil is a mixture of hydrocarbons.
A mixture means the substances are physically combined and not chemically bonded together.
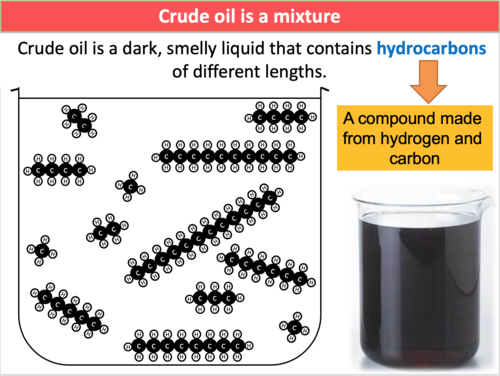
Each component in crude oil is a hydrocarbon (contains only carbon and hydrogen).
Why Crude Oil Is a Mixture
- Contains hydrocarbons of different chain lengths.
- Contains molecules with different boiling points.
- Can be separated by physical processes (fractional distillation).
Examples of Hydrocarbons in Crude Oil
Methane: \( \mathrm{CH_4} \)
Pentane: \( \mathrm{C_5H_{12}} \)
Decane: \( \mathrm{C_{10}H_{22}} \)
All of these contain only carbon and hydrogen.
Key Properties of Crude Oil
- Dark, viscous liquid.
- Formed from fossil fuels over millions of years.
- Non-renewable resource.
- Main source of fuels and petrochemicals.
| Feature | Crude Oil |
|---|---|
| Type | Mixture |
| Components | Hydrocarbons |
| Separation method | Fractional distillation |
Exam Insight
Do not describe crude oil as a compound.
It is not a single substance, it is a mixture of many hydrocarbons.
Example 1 (Conceptual):
Why is crude oil not a pure substance?
▶️ Answer/Explanation
Because it contains many different hydrocarbons.
They are not chemically bonded together.
Example 2 (Application):
Which of the following is found in crude oil?
A. Water B. Sodium chloride C. Hydrocarbons D. Oxygen gas
▶️ Answer/Explanation
Correct answer: C
Crude oil is a mixture of hydrocarbons.
Example 3 (Hard):
Explain fully why crude oil is described as a mixture of hydrocarbons.
▶️ Answer/Explanation
Crude oil contains many different molecules.
Each molecule contains only carbon and hydrogen.
The molecules have different chain lengths and boiling points.
They are physically mixed, not chemically bonded.
Therefore crude oil is a mixture of hydrocarbons.
