Edexcel iGCSE Chemistry -4.8 Fractional Distillation of Crude Oil- Study Notes- New Syllabus
Edexcel iGCSE Chemistry -4.8 Fractional Distillation of Crude Oil- Study Notes- New syllabus
Edexcel iGCSE Chemistry -4.8 Fractional Distillation of Crude Oil- Study Notes -Edexcel iGCSE Chemistry – per latest Syllabus.
Key Concepts:
4.8 describe how the industrial process of fractional distillation separates crude oil into fractions
4.8 Fractional Distillation of Crude Oil
Crude oil is a mixture of hydrocarbons with different boiling points.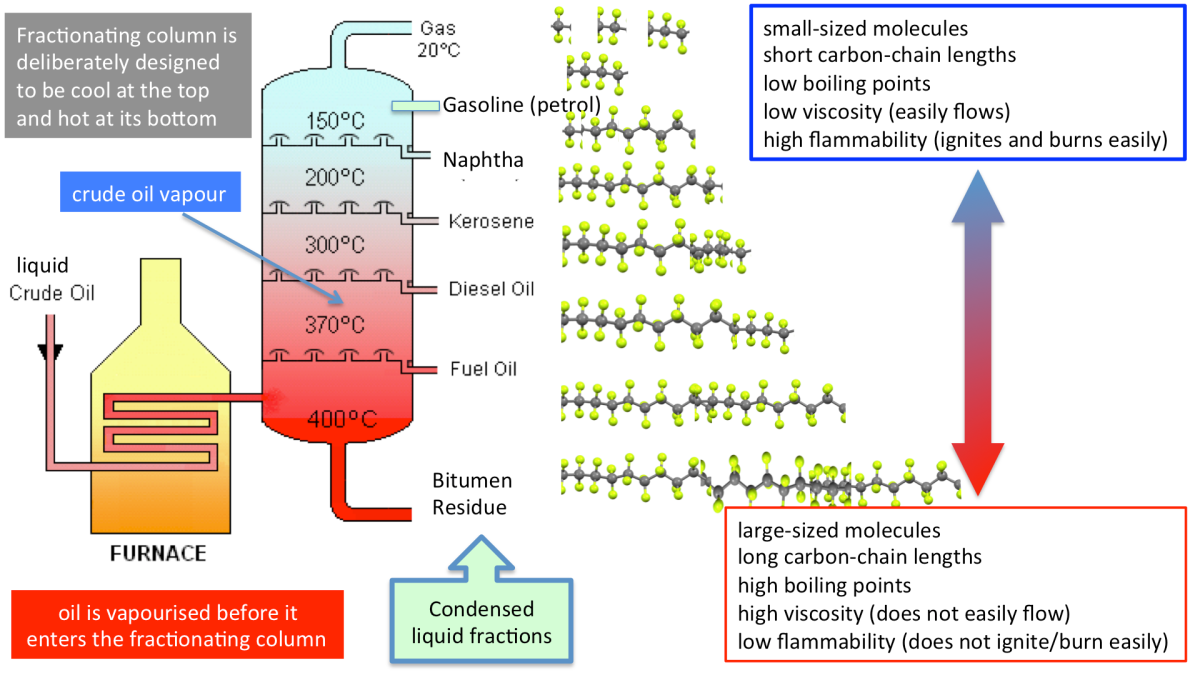
It is separated into useful products by fractional distillation.
This is a physical separation process, not a chemical reaction.
Principle
Hydrocarbons have different boiling points.
Smaller molecules:
- Lower boiling points
- Weaker intermolecular forces
- Rise higher in the column
Larger molecules:
- Higher boiling points
- Stronger intermolecular forces
- Condense lower in the column
Industrial Process
- Crude oil is heated in a furnace until most of it vaporises.
- The hot vapour enters the fractionating column.
- The column is hot at the bottom and cooler at the top.
- Vapours rise up the column.
- Each hydrocarbon condenses at the level where the temperature is just below its boiling point.
- The condensed liquids are drawn off as fractions.
Temperature Gradient
Bottom of column → highest temperature Top of column → lowest temperature
| Fraction | Boiling Point | Position in Column | Uses |
|---|---|---|---|
| Refinery gases | Lowest | Top | Heating, cooking |
| Petrol | Low | Upper middle | Fuel for cars |
| Kerosene | Medium | Middle | Jet fuel |
| Diesel | High | Lower middle | Fuel for vehicles |
| Bitumen | Highest | Bottom | Road surfacing |
Why It Works
Each fraction contains hydrocarbons with similar chain lengths and similar boiling points.
They condense at similar temperatures and are collected together.
Example 1 (Conceptual):
Why do smaller hydrocarbons rise higher up the column?
▶️ Answer/Explanation
They have lower boiling points.
They condense at lower temperatures near the top of the column.
Example 2 (Application):
Which fraction would you expect to have the highest boiling point?
▶️ Answer/Explanation
Bitumen.
It contains very long hydrocarbon chains.
Example 3 (Hard):
Explain fully how fractional distillation separates crude oil into fractions.
▶️ Answer/Explanation
Crude oil is heated until it vaporises.
The vapour enters a fractionating column that is hot at the bottom and cooler at the top.
Different hydrocarbons have different boiling points.
As vapours rise, they cool.
Each hydrocarbon condenses at the temperature just below its boiling point.
Long-chain hydrocarbons condense low in the column.
Short-chain hydrocarbons condense high in the column.
The liquids are collected as separate fractions.
The process is physical separation based on boiling point differences.
