1.2 Uncertainties and errors
Essential Idea:
Scientists aim towards designing experiments that can give a “true value” from their measurements, but due to the limited precision in measuring devices,
they often quote their results with some form of uncertainty.
Understandings:
- Random and systematic errors
- Absolute, fractional and percentage uncertainties
- Error bars
- Uncertainty of gradient and intercepts
Applications and Skills:
- Explaining how random and systematic errors can be identified and reduced
- Collecting data that include absolute and/or fractional uncertainties and stating these as an uncertainty range (expressed as: best estimate ±
uncertainty range) - Propagating uncertainties through calculations involving addition, subtraction, multiplication, division and raising to a power
- Determining the uncertainty in gradients and intercepts
PHYSICAL WORLD
WHAT IS SCIENCE?
Purpose of science is to produce useful models of reality. Most scientific investigations use some form of the scientific method.
Science as defined above, is sometimes called pure science to differentiate it from applied science, which is the application of research to human needs. Fields of science are commonly divided into two major categories.
- Natural science : The science in which we study about the natural world. Natural science includes physics, chemistry, biology, etc.
- Social science : It is the systematic study of human behavior and society.
WHAT IS PHYSICS?
There are two main thrusts in physics :
- Unification : In physics, attempt is made to explain various physical phenomena in terms of just few concepts and laws. Attempts are being made to unify fundamental forces of nature in the pursuit of unification.
- Reductionism : Another attempt made in physics is to explain a macroscopic system in terms of its microscopic constituents. This pursuit is called reductionism.
Keep in memory
- Information received through the senses is called observation.
- An idea that may explain a number of observations is called hypothesis.
- A hypothesis that has been tested many times is called scientific theory.
- A scientific theory that has been tested and has always proved true is called scientific law.
SCOPE AND EXCITEMENT OF PHYSICS
Classical physics deals mainly with macroscopic phenomena consisting of the study of heat, light, electricity, magnetism, optics, acoustics, and mechanics. Since the turn of the 20th century, the study of quantum mechanics and relativity has become more important. Today, physics is often divided into branches such as nuclear physics, particle physics, quantum physics, theoretical physics, and solid-state physics. The study of the planets, stars, and their interactions is known as astrophysics, the physics of the Earth is called geophysics, and the study of the physical laws relating to living organisms is called biophysics.
Physics is exciting in many ways. Application and exploitation of physical laws to make useful devices is the most interesting and exciting part and requires great ingenuity and persistence of effort.
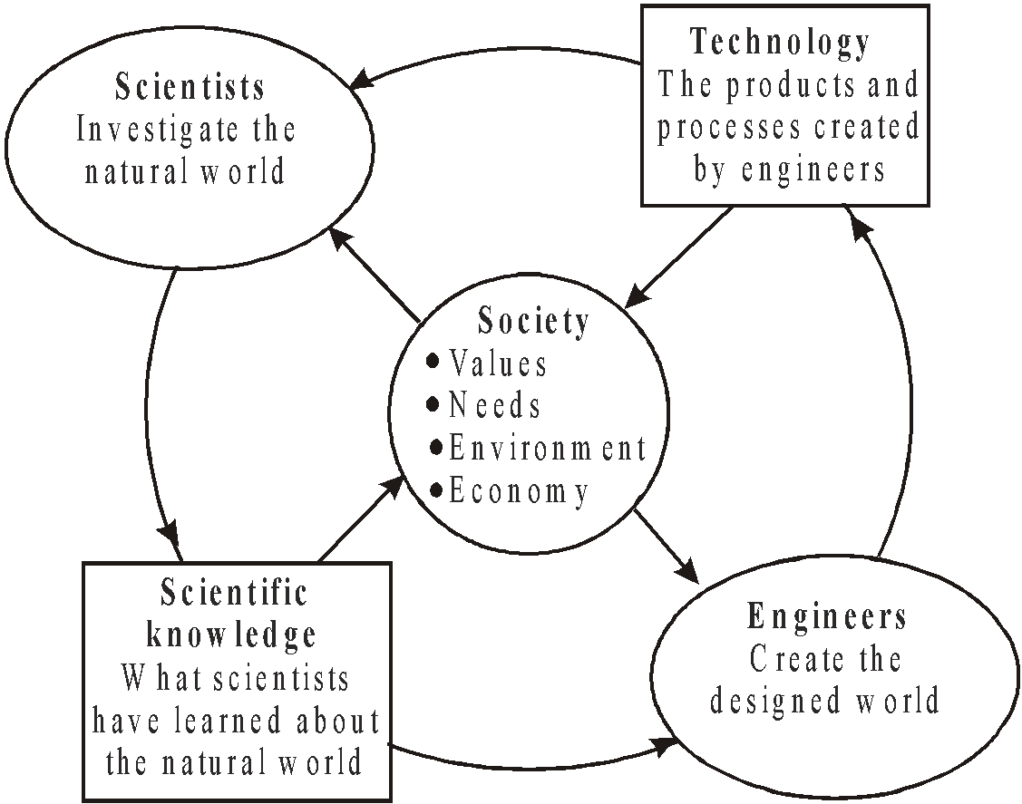
PHYSICS, TECHNOLOGY AND SOCIETY
SCIENCE
- A body of knowledge
- Seeks to describe and understand the natural world and its physical properties
- Scientific knowledge can be used to make predictions
- Science uses a process–the scientific method–to generate knowledge
ENGINEERING
- Design under constraint
- Seeks solutions for societal problems and needs
- Aims to produce the best solution from given resources and constraints
- Engineering uses a process–the engineering design process–to produce solutions and technologies
TECHNOLOGY
- The body of knowledge, processes and artifacts that result from engineering
- Almost everything made by humans to solve a need is a technology
- Examples of technology include pencils, shoes, cell phones, and processes to treat water
LINK BETWEEN TECHNOLOGY AND PHYSICS
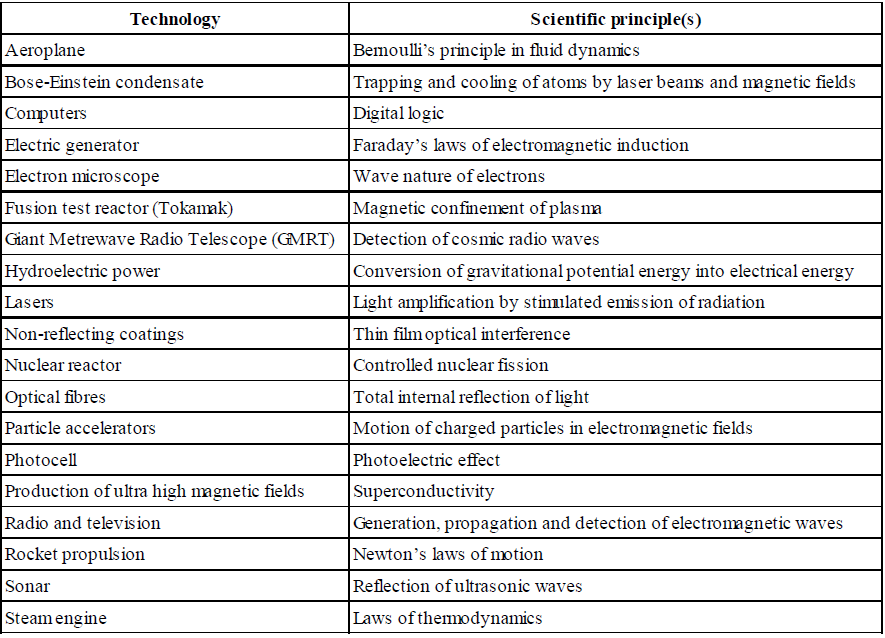
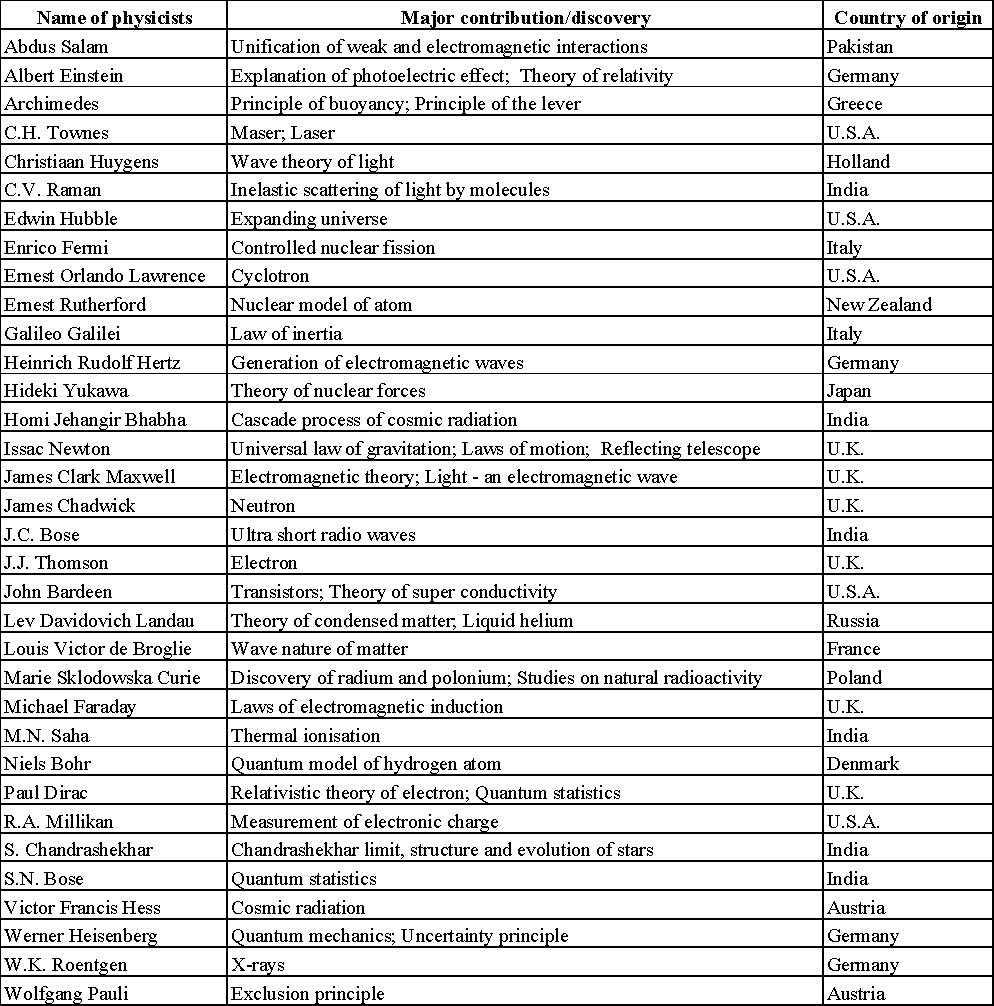
DOES IMAGINATION PLAY ANY ROLE IN PHYSICS?
HOW IS SCIENCE DIFFERENT FROM TECHNOLOGY?
FUNDAMENTAL FORCES IN NATURE
- Gravitational force
- Weak nuclear force
- Electromagnetic force
- Strong nuclear force
FUNDAMENTAL FORCES OF NATURE
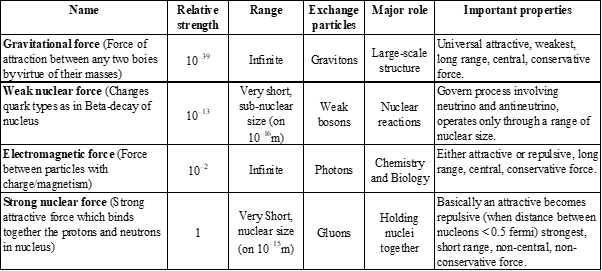
- Theories that postulate the unification of the strong, weak, and electromagnetic forces are called Grand Unified Theories (often known by the acronym GUTs).
- Theories that add gravity to the mix and try to unify all four fundamental forces into a single force are called Super Unified Theories.
- Theory that describes the unified electromagnetic and weak interactions is called the Standard Electroweak Theory, or sometimes just the Standard Model.
PROGRESS IN UNIFICATION OF DIFFERENT FORCES/DOMAINS IN NATURE

WHAT IS ELECTROMAGNETIC FORCE?
NATURE OF PHYSICAL LAWS
UNITS AND MEASUREMENTS
PHYSICAL QUANTITY
TYPES OF PHYSICAL QUANTITY
- Fundamental quantities : The physical quantities which do not depend upon other physical quantities are called fundamental or base physical quantities. e.g. mass, length, time temperature electric current, luminous intensity and amount of substance.
- Derived quantities : The physical quantities which depend on fundamental quantities are called derived quantities
e.g. speed, acceleration, force, etc.
UNIT
- It should be suitable in size (suitable to use)
- It should be accurately defined (so that everybody understands the unit in same way)
- It should be easily reproducible.
- It should not change with time.
- It should not change with change in physical conditions i.e., temperature, pressure, moisture etc.
- It should be universally acceptable.

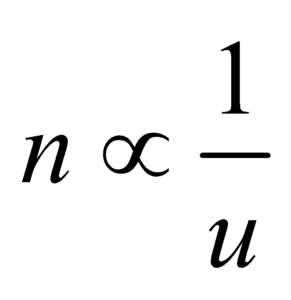
FUNDAMENTAL (OR BASE) AND DERIVED UNITS
SYSTEM OF UNITS
DIFFERENT TYPES OF SYSTEM OF UNITS
C.G.S. (Centimetre – Gram – Second) system. (Gaussian system of units): In this system the unit of length is centimetre, mass is gram and time is second.
M.K.S (Metre – Kilogram – Second) system. This system is related to mechanics only. In this system the unit of length is metre, mass is kilogram and time is second.
S.I. (International system) units. (Introduced in 1971) Different countries use different set of units. To avoid complexity, by international agreement, seven physical quantities have been chosen as fundamental or base physical quantities and two as supplementary. These quantities are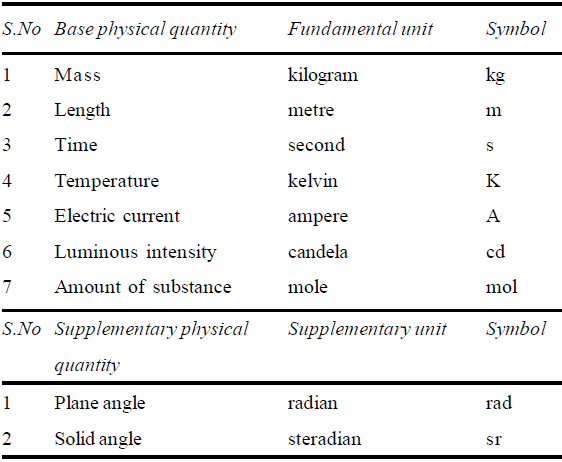
MERITS OF S.I. UNITS
- SI is a coherent system of units: This means that all derived units are obtained by multiplication and division without introducing any numerical factor.
- SI is a rational system of units: This is because it assigns only one unit to a particular physical quantity.
- SI is an absolute system of units: There is no gravitational unit in this system.
- SI system is applicable to all branches of science.
CONVENTIONS OF WRITING OF UNITS AND THEIR SYMBOLS
- Unit is never written with capital initial letter.
- For a unit named after scientist the symbol is a capital letter otherwise not.
- The unit or symbol is never written in plural form.
- Punctuations marks are not written after the symbol.
DEFINITIONS OF FUNDAMENTAL UNITS
- Metre : One metre is equal to 1650763.73 wavelength in vacuum of the radiation corresponding to transition between the levels 2p10 and 5d5 of the krypton – 86 atom
The distance travelled by light in vacuum in
- Kilogram : The mass of cylinder (of height and diameter 39 cm) made of Platinum-iridium alloy kept at International Bureau of weights and measures in paris is defined as 1kg.
- Second : It is the duration of 9,192,631,770 periods of radiation corresponding to the transition between the two hyperfine levels of the ground state of Caesium (133) atom.
- Ampere : It is the current which when flows through two infinitely long straight conductors of negligible cross-section placed at a distance of one metre in air or vacuum produces a force of 2 × 10–7 N/m between them.
- Candela : It is the luminous intensity in a perpendicular direction, of a surface of 1/600,000 square metre of a black body at the temperature of freezing platinum under a pressure of 1.013 × 105 N/m2.
- Kelvin : It is the 1/273.16 part of thermodynamic temperature of triple point of water.
- Mole : It is the amount of substance which contains as many elementary entities as there are in 0.012 kg of Carbon-12.
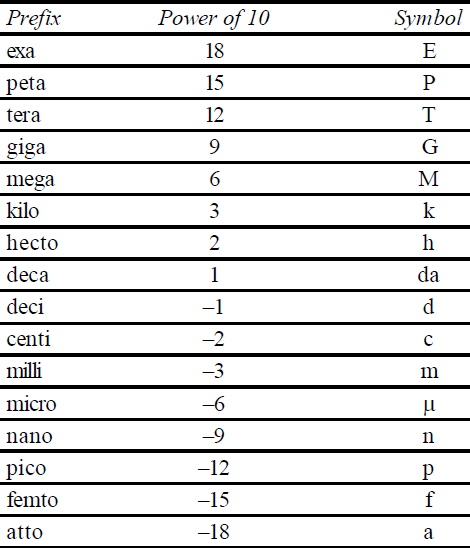
S.I. PREFIXES
SOME IMPORTANT PRACTICAL UNITS
- For large distance (macro-cosm)
1 A.U. = 1.496 × 1011m
1 ly = 9.46 × 1015m
1 parsec = 3.1 × 1016m
- For small distance (micro-cosm)
1 nanometre = 10–9m
1 angstorm = 10–10m
1 fermi = 10–15m
- For small area
1 barn = 10–28m2 - For heavy mass
1 ton = 1000kg
1 quintal = 100kg
1 slug = 14.57kg
1 C.S.L (chandrasekhar limit) = 1.4 times the mass of the sun - For small mass
1 amu = 1.67 x 10–27kg
1 pound = 453.6g = 0.4536 kg - For small time
1 shake = 10–8s - For large time
- For measuring pressure
DIMENSIONS
DIMENSIONAL FORMULA
DIMENSIONAL EQUATION
CLASSIFICATION OF PHYSICAL QUANTITIES (ON THE BASIS OF DIMENSIONS)

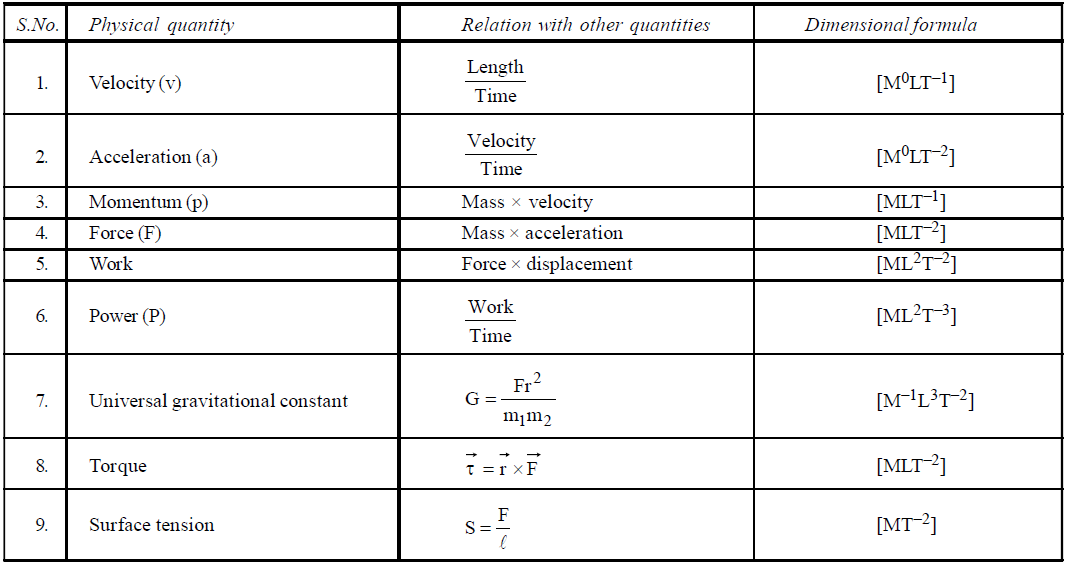
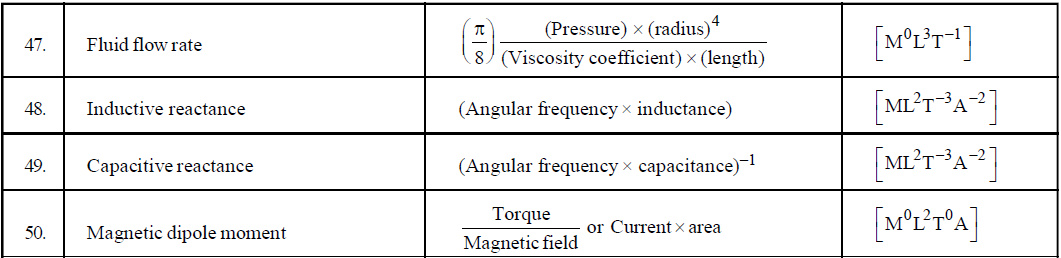
DIMENSIONAL FORMULA OF SOME IMPORTANT PHYSICAL QUANTITIES

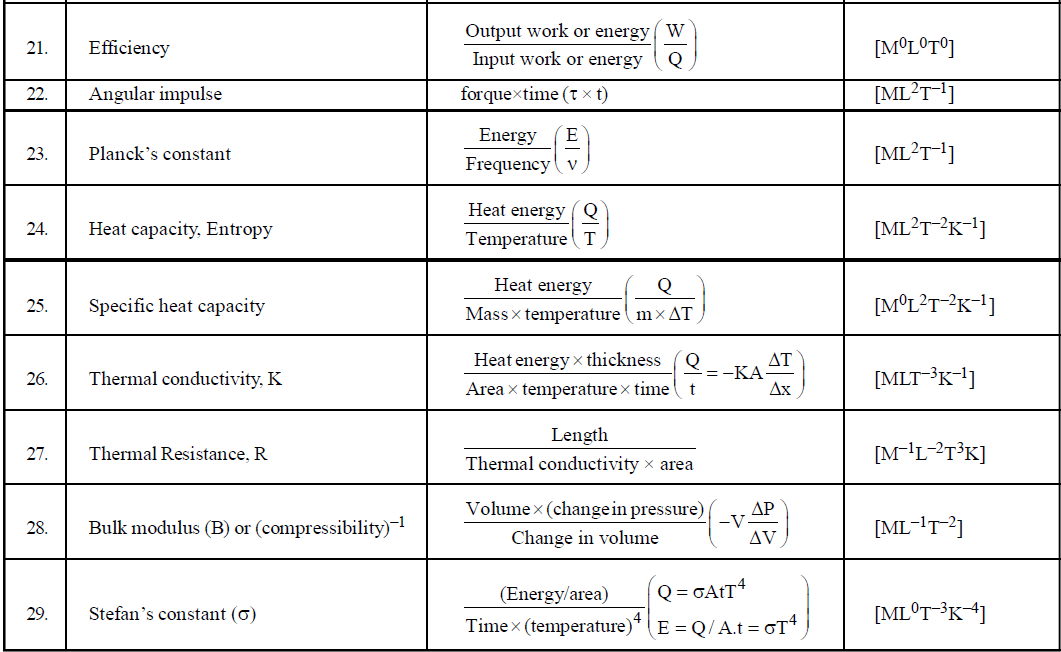


- To find dimensions of a typical physical quantity which is involved in a number of formulae, try to use that formula which is easiest for you. For example if you want to find the dimensional formula of magnetic induction then you can use the following formulae
- If you have to find the dimensional formula of a combination of physical quantities, then instead of finding the dimensional formula of each, try to correlate the combination of physical quantities with a standard formula. For example, if you have to find the dimension of CV2, then try to use formula
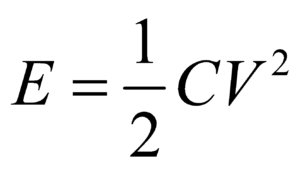 where E is energy of a capacitor.
where E is energy of a capacitor. 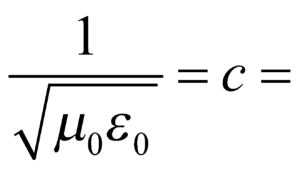 velocity of light in vacuum
velocity of light in vacuum- Dimensions of the following are same
- Dimensions of the following are same
- The dimension of RC =
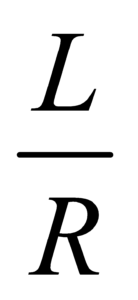 is same as that of time
is same as that of time - Dimensions of the following are same
- Dimensions of the following are same
- Dimensions of the following are same
- Dimensions of the following are same
- Dimensions of the following are same
KEEP IN MEMORY
- all trigonometric ratio is [M0L0T0]
- x in ex is [M0L0T0]
- ex is [M0L0T0]
- x in log x is [M0L0T0]
- log x is [M0L0T0]
DIMENSIONAL ANALYSIS AND ITS APPLICATIONS
USES OF DIMENSIONS
- Conversion of one system of unit into another
- Checking the accuracy of various formulae
- Derivation of formula
LIMITATIONS OF DIMENSIONAL ANALYSIS
- No information about the dimensionless constant is obtained during dimensional analysis
- Formula cannot be found if a physical quantity is dependent on more than three physical quantities.
- Formula containing trigonometrical /exponential function cannot be found.
- If an equation is dimensionally correct it may or may not be absolutely correct.
SIGNIFICANT FIGURES
RULES TO DETERMINE THE NUMBERS OF SIGNIFICANT FIGURES
- All non-zero digits are significant. 235.75 has five significant figures.
- All zeroes between two non-zero digits are significant. 2016.008 has seven significant figures.
- All zeroes occurring between the decimal point and the non-zero digits are not significant provided there is only a zero to left of the decimal point. 0.00652 has three significant figures.
- All zeroes written to the right of a non-zero digit in a number written without a decimal point are not significant. This rule does not work if zero is a result of measurement. 54000 has two significant figures whereas 54000m has five significant figures.
- All zeroes occurring to the right of a non-zero digit in a number written with a decimal point are significant. 32.2000 has six significant figures.
- When a number is written in the exponential form, the exponential term does not contribute towards the significant figures. 2.465 × 105 has four significant figures.
KEEP IN MEMORY
- The significant figures depend upon the least count of the instrument.
- The number of significant figure does not depend on the units chosen.
ROUNDING OFF
- If digit to be dropped is less than 5 then preceding digit should be left unchanged.
- If digit to be dropped is more than 5 then one should raise preceding digit by one.
- If the digit to be dropped is 5 followed by a digit other than zero then the preceding digit is increased by one.
- If the digit to be dropped is 5 then the preceding digit is not changed if it is even.
- If digit to be dropped is 5 then the preceding digit is increased by one if it is odd.
ARITHMETICAL OPERATIONS WITH SIGNIFICANT FIGURES AND ROUNDING OFF
- For addition or subtraction, write the numbers one below the other with all the decimal points in one line. Now locate the first column from the left that has a doubtful digit. All digits right to this column are dropped from all the numbers and rounding is done to this column. Addition subtraction is then done.
Step-2 :- 23.6 + 8.7=32.3
- In multiplication and division of two or more quantities, the number of significant digits in the answer is equal to the number of significant digits in the quantity, which has minimum number of significant digits.
ACCURACY, PRECISION OF INSTRUMENTS AND ERRORS IN MEASUREMENTS
The following diagrams illustrate the meaning of terms accuracy and precision
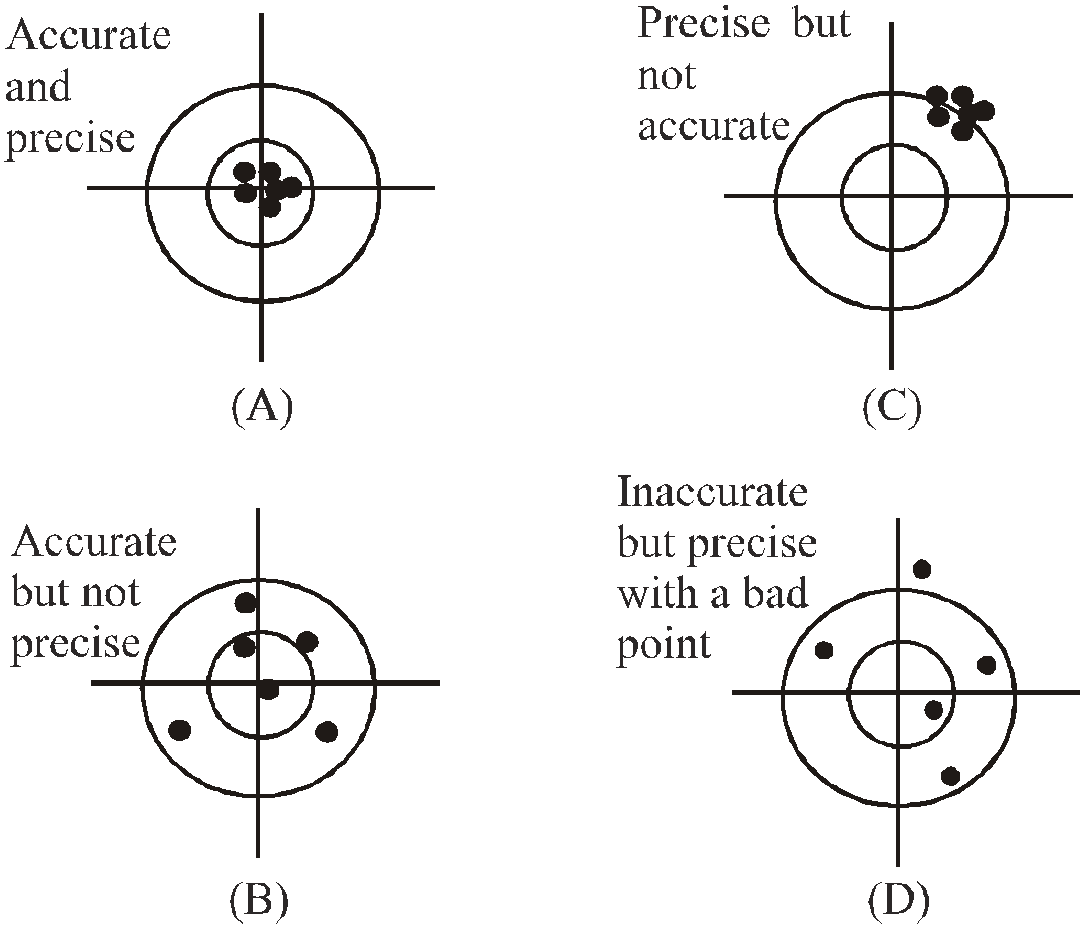
- Accuracy depends on the least count of the instrument used for measurement.
- In the addition and subtraction operation, the result contains the minimum number of decimal places of the figures being used
- In the multiplication and division operation, the result contains the minimum number of significant figures.
- Least count (L.C.) of vernier callipers = one MSD – one VSD
- Least count of screw gauge (or spherometer)

where pitch is the ratio of number of divisions moved on linear scale and number of rotations given to circular scale. - Pure number or unmeasured value do not have significant numbers
- Change in the position of decimal does not change the number of significant figures. Similarly the change in the units of measured value does not change the significant figures.
COMMON ERRORS IN MEASUREMENTS
EXPERIMENTAL ERRORS
- by the lack of perfection of observer
- if the measuring instrument is not perfectly sensitive.
- measuring instrument having a zero error.
- an instrument being incorrectly calibrated (such as slow- running-stop clock)
- the observer persistently carrying out a mistimed action (e.g., in starting and stopping a clock)
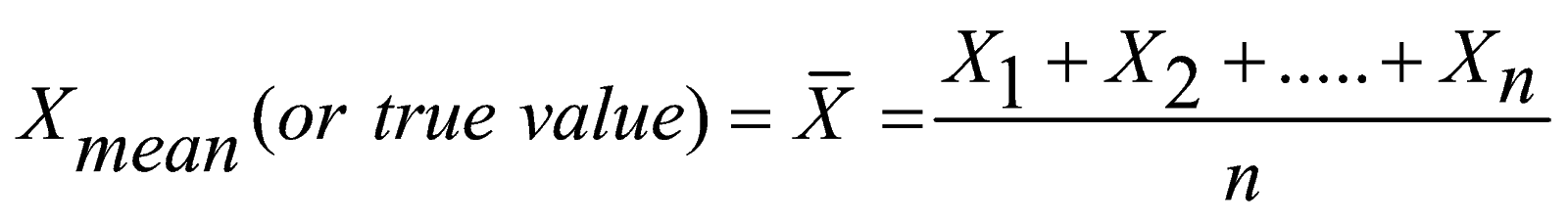
METHODS OF EXPRESSING ERROR
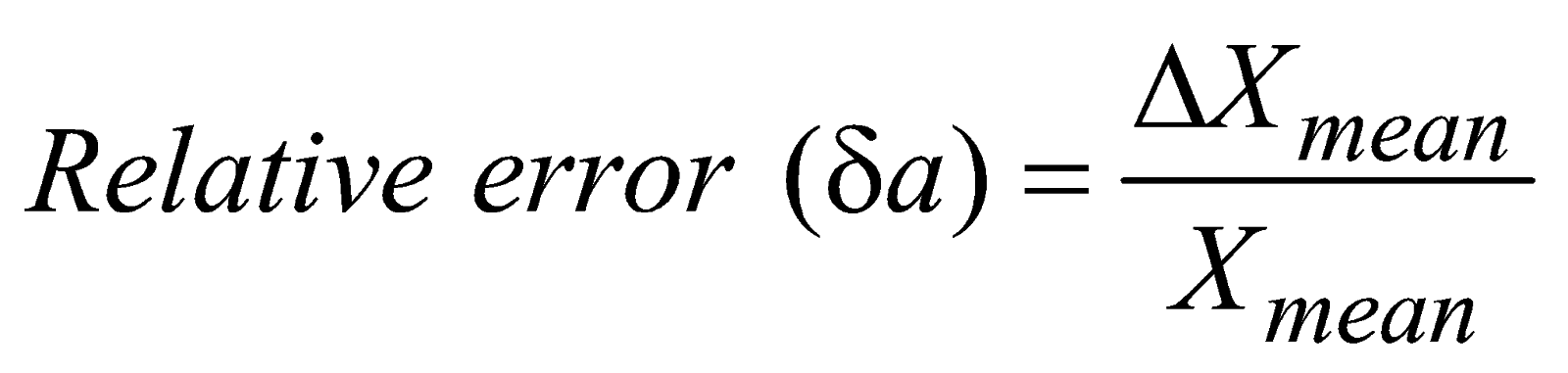
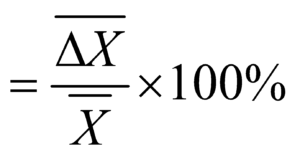
TO FIND THE MAXIMUM ERROR IN COMPOUND QUANTITIES
We have to find the sum or difference of two values given as (a ± Δa) and (b ± Δb), we do it as follows
PRODUCT AND QUOTIENT
We add the fractional or percentage errors in case of finding product or quotient.
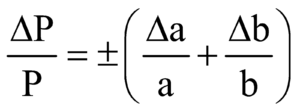
If x = an then

- More the accuracy, smaller is the error.
- Absolute error |ΔX| is always positive.
- |ΔX| has the same dimensions as that of X.
- If the least count of measuring instrument is not given and the measured value is given the least error in the measurement can be found by taking the last digit to be 1 and rest digit to be zero. For e.g. if the measured value of mass m = 2.03 kg then
.
- If a number of physical quantities are involved in an expression then the one with higher power contributes more in errors and therefore should be measured more accurately.
- Relative error is a dimensionless quantity.
- We are always interested in calculating the maximum possible error.
