iGCSE Physics (0625) 5.2.4 Half-life -Exam Style Questions Paper 1 - New Syllabus
Question
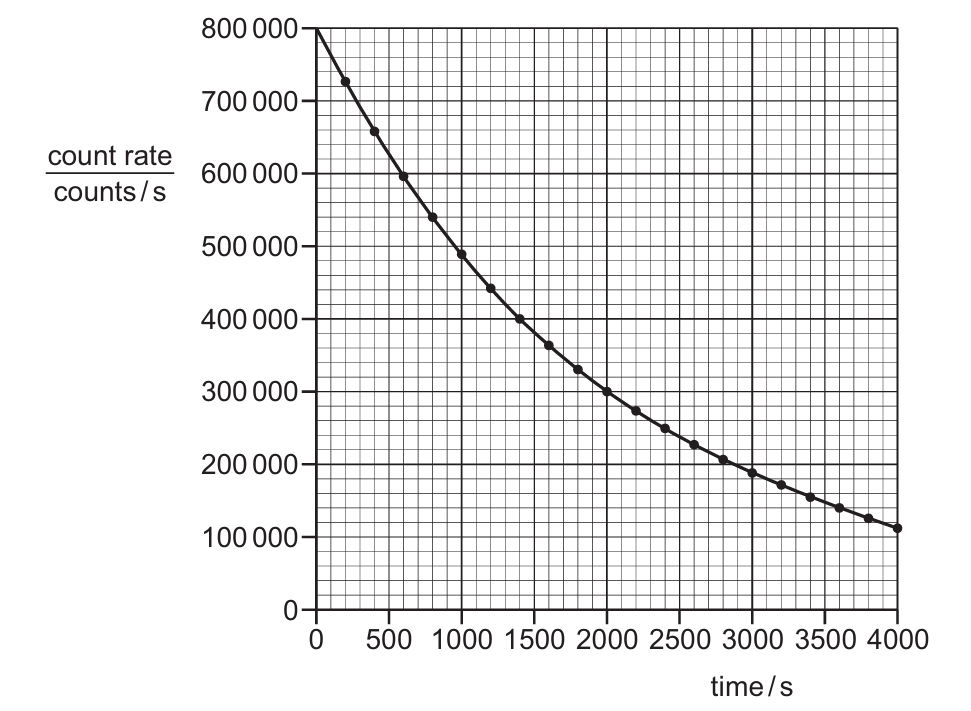
▶️ Answer/Explanation
Detailed solution:
The half-life is the time taken for the activity (count rate) of a radioactive sample to decrease to half of its initial value. From the graph, the initial count rate at $t = 0$ s is $800~000$ counts/s. Half of this initial value is $\frac{800~000}{2} = 400~000$ counts/s. By locating $400~000$ counts/s on the y-axis and moving horizontally to the curve, we drop down to the x-axis to find the corresponding time. The intersection occurs at approximately $1400$ s. Therefore, the approximate half-life is $1400$ s.
Question
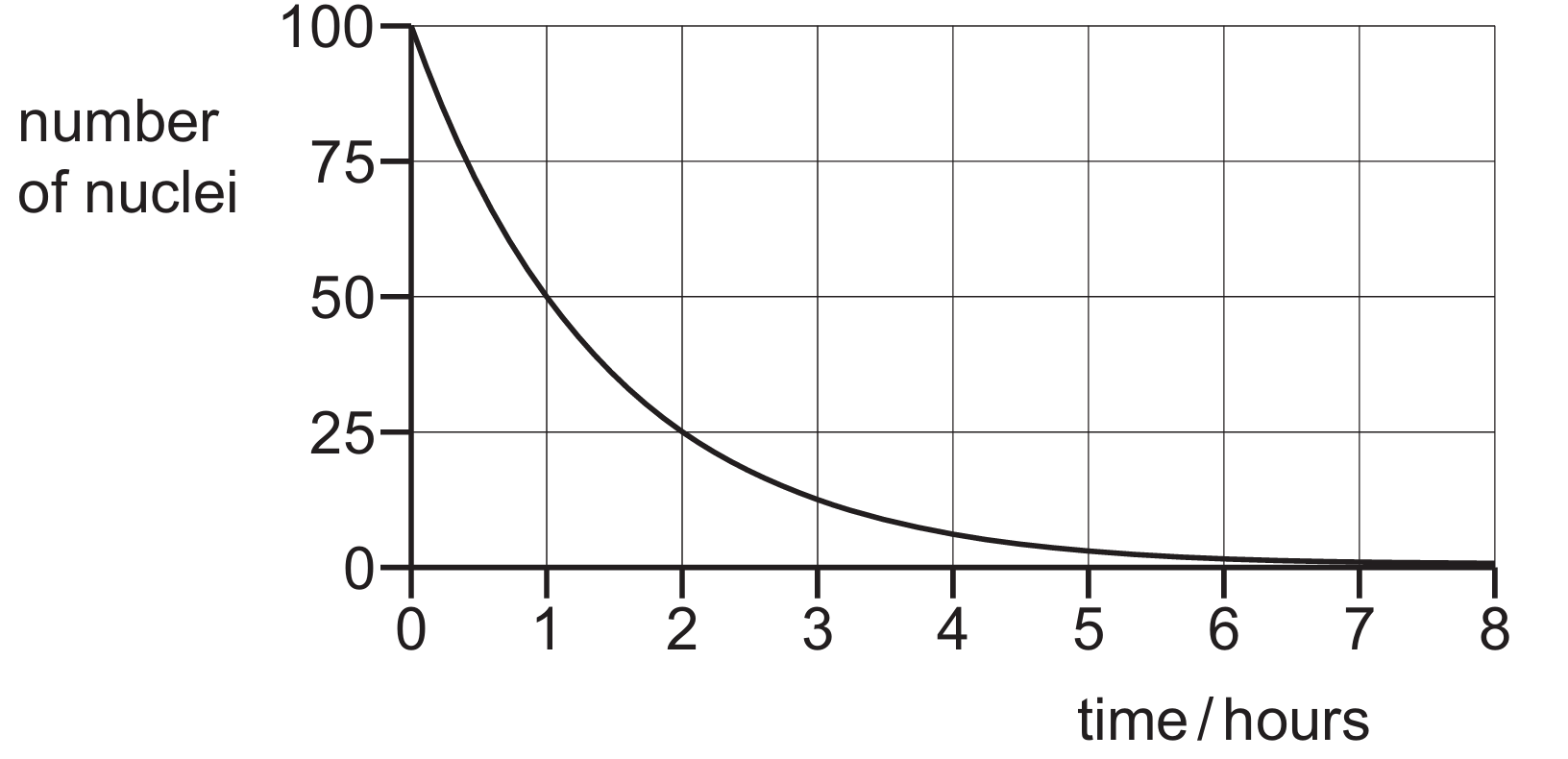
▶️ Answer/Explanation
Detailed solution:
The half-life is defined as the time taken for half the nuclei in a sample to decay. Starting with $100$ nuclei at $t = 0$, half of the initial amount is $\frac{100}{2} = 50$ nuclei. By locating $50$ on the vertical axis (number of nuclei) and moving horizontally to the curve, we drop down to the horizontal axis to find the corresponding time. The graph shows that the sample reaches $50$ nuclei at $t = 1$ hour. Similarly, it takes another hour to halve again to $25$ nuclei at $t = 2$ hours, confirming the half-life is $1$ hour.
Question
An isotope of strontium decays by $\beta$ emission. It takes $87$ hours for its activity to fall to $\frac{1}{8}$ of its original value.
What is the half-life of the isotope?
▶️ Answer/Explanation
Detailed solution:
The activity falls to $\frac{1}{8}$ of its original value. Since $(\frac{1}{2})^{3} = \frac{1}{8}$, this represents exactly $3$ half-lives.
The total time taken for this decay is given as $T_{total} = 87$ hours.
To find the duration of one half-life ($t_{1/2}$), we divide the total time by the number of half-lives: $t_{1/2} = \frac{87}{3}$.
Performing the calculation gives $t_{1/2} = 29$ hours.
Therefore, the half-life of the strontium isotope is $29$ hours, which matches option B.
