Question
What is the maximum mass, in g, of magnesium oxide that can be obtained from the reaction of oxygen with 2.4 g of magnesium?
A. 2.4
B. 3.0
C. 4.0
D. 5.6
▶️Answer/Explanation
C
\[{\text{2Mg(s)}} + {{\text{O}}_2}({\text{g)}} \to {\text{2Mg}}{{\text{O}}}({\text{g)}}\]
Given, 2.4 g of Mg
Number of moles of Mg = (Mass of Mg) ÷ (Molar mass of Mg)
i.e. Number of moles of Mg = 2.4/24 = 0.1 moles of Mg
From the equation above, 2 moles of Mg gives 2 moles of MgO.
Hence, 0.1 mole of Mg will give 0.1 mole of MgO.
Maximum mass of MgO that can be obtained = 0.1×(Molecular mass of MgO) = 0.1×(24+16) = 4.0 g
Question
\({\text{5 d}}{{\text{m}}^{\text{3}}}\) of carbon monoxide, CO(g), and \({\text{2 d}}{{\text{m}}^{\text{3}}}\) of oxygen, \({{\text{O}}_{\text{2}}}{\text{(g)}}\), at the same temperature and pressure are mixed together. Assuming complete reaction according to the equation given, what is the maximum volume of carbon dioxide, \({\text{C}}{{\text{O}}_{\text{2}}}{\text{(g)}}\), in \({\text{d}}{{\text{m}}^{\text{3}}}\), that can be formed?
\[{\text{2CO(g)}} + {{\text{O}}_2}({\text{g)}} \to {\text{2C}}{{\text{O}}_2}({\text{g)}}\]
A. 3
B. 4
C. 5
D. 7
▶️Answer/Explanation
B
According to Gay Lussac’s law:
2CO(g) + O2(g) → 2CO2(g)
2 vol 1 vol 2 vol
1 vol 0.5 vol 1 vol
1 vol of CO requires 0.5 vol of oxygen.
5 dm3 CO requires 2.5 dm3 of oxygen. Here, we have only 2 dm3 of oxygen. Here, oxygen is the limiting reagent.
0.5 vol of oxygen will form 1 vol of CO2
So, 2 dm3 of oxygen will form 4 dm3 of CO2.
Question
The volume of an ideal gas at 27.0 °C is increased from \({\text{3.00 d}}{{\text{m}}^{\text{3}}}\) to \({\text{6.00 d}}{{\text{m}}^{\text{3}}}\). At what temperature, in °C, will the gas have the original pressure?
A. 13.5
B. 54.0
C. 327
D. 600
▶️Answer/Explanation
C
Using ideal gas equation, we have
\(\frac{P1V1}{T1} = \frac{P2V2}{T2}\) where P=Pressure, V=Volume and T=Absolute temperature of the gas.
Given, P1=P2=P(say) (Since pressure is constant)
V1=V(say), where V is the initial volume of the gas.
V1 = 3 dm3 , V2 = 6 dm3
Therefore, V2=2V since final volume is double the initial volume.
T1=(27+273)K=300K
T2=?
Substituting the values obtained in the gas equation, we have
\(\frac{PV}{300} = \frac{P(2V)}{T2}\)
Solving, we get T2=final temperature of the gas=600K or (600–273)degree C or 327°C.
Question
5.0mol of Fe2O3(s) and 6.0mol of CO(g) react according to the equation below. What is the limiting reactant and how many moles of the excess reactant remain unreacted?
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2(g)

▶️Answer/Explanation
B
Fe2O3(s) + 3CO(g) → 2Fe(s) + 3CO2(g)
1 : 3
5 mole Fe2O3 requires 15 moles of CO.
We have only 6 moles of CO. Hence, CO is the limiting reactant. Fe2O3 is the excess reactant.
3 moles of CO require 1 mole of Fe2O3
So, 6 moles of CO will require 2 mole of Fe2O3.
Hence, moles of excess reactant remaining = 5 – 2 =3.
Question
Which electron transition emits energy of the longest wavelength?
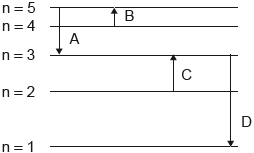
▶️Answer/Explanation
A
Wavelength is given by :
\(\frac{1}{\lambda}=RZ^2(\frac{1}{n_2^2} – \frac{1}{n_1^2})\)
Where λ = wavelength, R = Rydberg constant, Z = atomic number and n1 and n2 are principle quantum number.
For A, n2 = 3, n1 = 5
\(\frac{1}{\lambda_1}=RZ^2(\frac{1}{3^2} – \frac{1}{5^2})\)
\(\frac{1}{\lambda_1}=RZ^2(\frac{1}{9} – \frac{1}{25})\)
\(\lambda _1 = \frac{14.0625}{RZ^2}\)
For B, n2 = 5, n1 = 4
In this case, energy emission is not possible since emission occurs only from higher n to lower n.
For C, n2 = 3, n1 = 2
In this case, energy emission is also not possible since emission occurs only from higher n to lower n.
For D, n2 = 1, n1 = 3
\(\frac{1}{\lambda_4}=RZ^2(\frac{1}{1^2} – \frac{1}{3^2})\)
\(\lambda _4 = \frac{1.125}{RZ^2}\)
Hence, energy of longest wavelength is emitted in case of electron A.
