Question
Which statement best describes the intramolecular bonding in HCN(l)?
A. Electrostatic attractions between \({{\text{H}}^ + }\) and \({\text{C}}{{\text{N}}^ – }\) ions
B. Only van der Waals’ forces
C. Van der Waals’ forces and hydrogen bonding
D. Electrostatic attractions between pairs of electrons and positively charged nuclei
▶️Answer/Explanation
D
HCN has Electrostatic attractions between pairs of electrons of \({\text{C}}{{\text{N}}^ – }\) ion and positively charged nuclei of \({{\text{H}}^ + }\) .
Question
Which molecule has the shortest bond between carbon atoms?
A. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\)
B. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}\)
C. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\)
D. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{C}}{{\text{l}}_{\text{2}}}\)
▶️Answer/Explanation
C
HC≡CH , C -C Bond order =3
H2C=CH2 , C-C Bond order =2
H3C-CH3 , C-C Bond order =1
ClH2C-CH2Cl , C-C Bond order =1
More the bond order, lesser will be the bond length.
Hence, \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\) has the shortest bond between carbon atoms.
Question
When \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\), \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}\) and \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\) are arranged in order of increasing carbon-carbon bond strength (weakest bond first), what is the correct order?
A. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}\)
B. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}\)
C. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\)
D. \({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}\)
▶️Answer/Explanation
C
HC≡CH , C -C Bond order =3
H2C=CH2 , C-C Bond order =2
H3C-CH3 , C-C Bond order =1
Higher the bond order, lower is the bond length and higher is the bond strength.
So, the increasing order of carbon-carbon bond strength is :
\({{\text{C}}_{\text{2}}}{{\text{H}}_{\text{6}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{4}}}{\text{, }}{{\text{C}}_{\text{2}}}{{\text{H}}_{\text{2}}}\)
Question
The number of electrons in the valence shell of elements A and B, are 6 and 7 respectively. What is the formula and type of bonding in a compound formed by these elements?
A. \({{\text{A}}_{\text{2}}}{\text{B}}\), covalent
B. \({\text{A}}{{\text{B}}_{\text{2}}}\), covalent
C. \({{\text{A}}_{\text{2}}}{\text{B}}\), ionic
D. \({\text{A}}{{\text{B}}_{\text{2}}}\), ionic
▶️Answer/Explanation
B
Each atom requires 8 electrons to complete its shell. Here, A is short of two electrons and B is short of 1 electron to complete its shell.
Two B atoms share its electrons with A forming two A-B covalent bonds.
Compound formed will be \({\text{A}}{{\text{B}}_{\text{2}}}\) and covalent.
Question
Which bonds are arranged in order of increasing polarity?
A. H–F \( < \) H–Cl \( < \) H–Br \( < \) H–I
B. H–I \( < \) H–Br \( < \) H–F \( < \) H–Cl
C. H–I \( < \) H–Br \( < \) H–Cl \( < \) H–F
D. H–Br \( < \) H–I \( < \) H–Cl \( < \) H–F
▶️Answer/Explanation
C
Order of the polarity of the molecules depends on the electronegativity of the bonded atoms. The E.N order for halogens is given below.
Therefore, the order of polarity of hydrogen halide molecules is
H-I<H-Br<H-Cl<H-F.
Question
Which single covalent bond is the most polar, given the following electronegativity values?
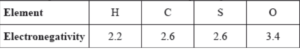
A. C–O
B. S–H
C. C–H
D. O–H
▶️Answer/Explanation
D
Polarity is determined by difference of electronegativity values of atoms.
E.N. difference : C-O = 3.4-2.6 = 0.8
E.N. difference : S-H = 2.6-2.2 = 0.4
E.N. difference : C-H = 2.6-2.2 = 0.4
E.N. difference : O-H = 3.4-2.2 = 1.2
Hence, O-H has maximum difference of electronegativity, it is most polar.
